| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Original Article
Volume 15, Number 1, February 2024, pages 47-55
Evaluation of the Existence of Post-COVID-19 Tachycardia in a Community Healthcare System
Jifeng Wanga, Dhaval Patela, Shane Robinsonb, e, Ania Rynarzewskac, Oluseyi Abidoyed
aInternal Medicine Department, Northeast Georgia Medical Center, GME, Gainesville, GA, USA
bGME Research, Northeast Georgia Medical Center, Gainesville, GA, USA
cGeorgia College and State University, Milledgeville, GA, USA
dHematology and Oncology, Mayo Clinic, GME, Pheonix, AZ, USA
eCorresponding Author: Shane Robinson, GME Research, Northeast Georgia Medical Center, Gainesville, GA, USA
Manuscript submitted December 12, 2023, accepted January 31, 2024, published online February 28, 2024
Short title: PCT in a Community Healthcare System
doi: https://doi.org/10.14740/cr1604
| Abstract | ▴Top |
Background: Post-coronavirus disease 2019 (COVID-19) syndrome derives from lingering symptoms after an acute COVID-19 infection. Palpitation was one of the most common symptoms of post-COVID-19 syndrome that correlated with objective data such as persisting sinus tachycardia; but to our best knowledge, there is a scarcity of research regarding the association of COVID-19 and sinus tachycardia in the post-acute setting. Therefore, the purpose was to identify if there is an association between COVID-19 infection and sinus tachycardia in the post-acute phase, namely post-COVID-19 tachycardia (PCT) other than inappropriate sinus tachycardia (IST) and postural orthostatic tachycardia syndrome (POTS).
Methods: This retrospective observational study entails 1,425 patients admitted for COVID-19 infection with the interest in finding an association with PCT. The prevalence of PCT was evaluated using descriptive statistics, predictions of patient characteristics and comorbidities were identified using multinomial logistic regression, and associations between patient comorbidities and characteristics were evaluated with corresponding Pearson Chi-square test and post hoc tests Phi and Cramer’s V.
Results: The percentage of patients with PCT in our sample of interest was an average of 28.18%. There was a strong association of PCT with patients of age group less than 65 years. Other clinical characteristics, such as shorter length of stay, unknown smoking status, and patients with commercial type insurance, had significant association with PCT. COVID-19 severity categorized as “less severe”, readmission rates within 30 days, and patients with less comorbidities were more likely to be associated with PCT.
Conclusions: PCT is likely a separate entity from IST and POTS, and an important entity under the umbrella of post-COVID-19 syndrome. It warrants further studies to elucidate the underlying pathophysiology and to confirm its presence as a distinct entity.
Keywords: COVID-19; Post-COVID-19 syndrome; Dysautonomia; Post-COVID-19 tachycardia
| Introduction | ▴Top |
Post-coronavirus disease 2019 (COVID-19) syndrome derives from lingering symptoms after COVID-19 infection [1]. These ongoing symptoms and complications continue to immerse and challenge practicing physicians in understanding and treating patient’s post-pandemic. Post-COVID-19 syndrome is poorly understood as it affects COVID-19 survivors with all levels of disease severity and can present in a variety of debilitating symptoms that affect the daily activities of living and their quality of life [1]. The official definition of post-COVID-19 syndrome is still lacking due to the novelty of this entity; however, literature refers to it as symptoms persisting for more than 3 weeks after the resolution of COVID-19 [2]. The United Kingdom National Institute of Health and Care Excellence (NICE) developed a classification system to more specifically define COVID syndrome that is grouped by the timeline of experienced symptoms from COVID-19 infection. The phases include “acute COVID-19” which involves symptoms lasting for up to 4 weeks, “ongoing symptomatic COVID-19” which is defined by experienced signs and symptoms from 4 to 12 weeks, and “post-COVID-19 syndrome” which classifies signs and symptoms of COVID-19 infection that are not clinically explained by an alternative diagnosis for more than 12 weeks from initial COVID-19 infection [3]. These groupings can make the timing and severity of post-COVID-19 syndrome more consistent, but the primary challenge is identifying the symptoms themselves.
While post-COVID-19 syndrome is being recognized globally with organization-provided guidelines by NICE, its diagnostic criteria lack universal agreement which hinders the full understanding and detection of post-COVID-19 syndrome [4, 5]. Patient symptoms for identifying post-COVID-19 syndrome derive from both subjective and objective measures that are used in the hope to help identify which symptoms have a potential relationship with post-COVID-19 syndrome. These subjectively reported symptoms include but are not limited to fatigue, muscle weakness, difficulty sleeping, dyspnea, hair loss, and palpitations [6]. The data on persistent symptomatology after the acute phase of this virus infection seems promising, but it is challenging to accurately diagnose post-COVID-19 syndrome with subjective data [7]. Many researchers are starting to gear the investigation from a subjective to an objective standpoint. Since dyspnea was the most common subjective data, association of this symptom to a cardiovascular etiology was speculated [7].
Palpitations were one of the most common symptoms that correlated with objective data such as persisting sinus tachycardia leading to the discovery of post-COVID-19 dysautonomia [4, 5, 8]. In a 6-month follow-up study, 9% of post-COVID-19 patients complained of palpitations which indicates a potential existence of a cardiac complication in post-COVID-19 syndrome [6]. Among national and international data, the most common post-COVID-19 symptom is fatigue or muscle weakness at 63% [9]. Sinus tachycardia is a normal compensatory cardiac rhythm that suggests fast heart beats at more than 100 beats per minute that is typically related to exertion or stress [10]. However, non-exertional sinus tachycardia, which is a heart rate of more than 100 beats per minute at rest, could be a concerning sign as it can be associated to different pathologies such as pulmonary emboli, hypoxia, cardiac arrythmias, sepsis, etc. [10]. Inappropriate sinus tachycardia (IST) and postural orthostatic tachycardia syndrome (POTS) were the two of the most common etiologies under post-COVID-19 dysautonomia researched according to the literature [11].
However, there have been several cases of post-COVID-19 tachycardia (PCT), which are not well established, and thought to be a separate entity from POTS and IST [8, 12]. This phenomenon differs from POTS due to elevated heart rates at rest and differs from IST due to asymptomatic persistent tachycardia and having no association to a primary cause [8, 13]. IST was found to be prevalent in patients with post-COVID-19 syndrome in a prospective study, and interestingly it was more prevalent in young women without comorbidities that experienced mild illness of COVID-19 infection as delineated by the National Institute of Health (NIH) guidelines of COVID-19 severity [8].
It is critical to determine the cause of sinus tachycardia as etiology can range from mild to life-threatening one [10]. Persistent sinus tachycardia that goes unrecognized can impact long-term health such as induced cardiomyopathy, myocardial infarctions, cardiac arrest, end-organ dysfunction, and among other threatening complications [14]. Therefore, our study’s purpose was to identify if there is an association between COVID-19 infection and sinus tachycardia in the post-acute phase excluding IST and POTS.
| Materials and Methods | ▴Top |
Subjects
Subjects within this retrospective chart review were adults aged 18 years and older that were admitted into Northeast Georgia Medical Center for COVID-19 from March 2020 to December 2021. All participants included had a diagnosis of sinus tachycardia with consecutive resting heart rate values of 100 beats per minute or greater during their admission. To further explain our sample selection, we have delineated definitions pertaining to COVID-19 infection, resolution of COVID-19 infection, and our inclusion/exclusion criteria.
Dataset was obtained through Northeast Georgia Health System (NGHS) by the Graduate Medical Education’s (GME) Research team.
The Institutional Review Board of Brenau University granted this study exemption from review. This study was carried out with ethical compliance with appropriate data governance and maintaining deidentified data through the Safe Harbor Method.
Inclusions and exclusions criterium
Our definition for resolution of COVID-19 infection is in part based on the Centers for Disease Control and Prevention (CDC) recommendations. Our criteria for ending isolation and precautions for people diagnosed with COVID-19 are listed in Table 1 [15]: having a negative repeat (second) COVID-19 test during the same admission with the first test being positive, more than or equal to 10 days after onset and resolution of fever for at least 24 h and improvement of symptoms, a documented formal diagnosis of post-COVID-19 syndrome, or the duration of 3 days prior to discharge or at day of discharge. Meeting at least one of these criteria will comply with the assumption of COVID-19 infection resolution.
 Click to view | Table 1. Definition of Resolution of COVID-19 Infection and Criteria for PCT |
We excluded a total number of 8,626 patients from complications that could have potentially caused an elevation in resting heart rate to ensure the study strictly focused on the association of tachycardia in post-COVID-19 syndrome. Our exclusion criteria consisted of the history of supraventricular tachycardia, atrial tachycardias, other arrythmias, ongoing sepsis, shock, venous thromboembolism, anxiety, taking beta-blockers or atrioventricular (AV) nodal blocking agents, extracorporeal membrane oxygenation, and critically ill patients or patients needing escalating levels of oxygen (NIH guidelines for COVID-19 severity).
It is plausible that our study was not able to include all the patients who would have otherwise met our inclusion criteria due to lack of a second COVID-19 test, shorter length of stay than our specified criteria or lack of documented post-COVID-19 syndrome diagnosis. We determined our best approach was to include data collected at the day of discharge or 3 days within discharge. We have also included data from 7 days within discharge to incorporate patients who were likely to be past their incubation periods. With this approach, we also aimed to address the two most common parameters associated with sinus tachycardia in the inpatient setting, hypoxia and fever. As a general rule, only safe discharges are tolerated in the United States’ current practice, therefore, a patient being safely discharged is presumably no longer hypoxic or requiring escalating levels of oxygen, or no longer having persistent fevers, which otherwise would warrant further investigations.
To define PCT, we needed to first ascertain that patients were free of COVID-19 infection based on CDC’s recommendations and secondly, to capture specific data that would associate elevated heart rates with no other conditions than the most recently infection of COVID-19. In this study, all patients with a negative repeat (from initial positive test from admission) COVID-19 test within 10 days before hospital discharge were defined as post-COVID-19 patients. To further classify patients with post-COVID-19 tachycardia, the resting heart rate was monitored over the 10 days before discharge, and all patients with consecutive resting heart rates over 100 beats per minute were diagnosed with PCT. To keep the focus of this study purely on PCT, patients with other underlying causes that could influence tachycardia were reviewed and excluded from the PCT diagnosed patients for this study. The following inclusion criterion (Table 1) was carefully selected to prove the latter: a documented formal diagnosis of sinus tachycardia, sinus tachycardia recorded between 7:00 pm to 6:00 am on 3 separate days, sinus tachycardia recorded between 12:00 am to 6:00 am on the day of discharge, or persistent sinus tachycardia on 3 separate occasions in a day for at least 3 days. Meeting these parameters helped us differentiate from POTS.
To set a clear difference between PCT and POTS, resting heart rate of > 100 beats per minute if any, must be recorded outside of physical therapy or mobility activity (e.g., 6-min walk test), in other words, they must be measured in a bedbound status. To measure this, heart rates from 7:00 pm to 6:00 am were recorded, given that physical therapy and ambulatory activities were not available during the nocturnal shift.
Outcomes
The primary observation was the prevalence of persistent sinus tachycardia after COVID-19 infection with the exclusion of other causes. Secondary observations consisted of the association of COVID-19 infection with clinical characteristics, comorbidities, COVID-19 infection severity, and readmission rates.
The demographics and clinical characteristics were body mass index, tobacco history, age, sex, race, length of stay, financial status, and severity of COVID-19. From a financial class standpoint, distinct groups of insurance coverage were studied: commercial, Medicare, Medicaid, self-pay and “other”. Smoking status was divided into the following: never smoked, former smoker, current smoker, and unknown status. Age groups consisted of less than 65, 65 - 75, and over 75.
Comorbidities of interest that were included in our study were type 2 diabetes mellitus, coronary artery disease, congestive heart disease, chronic obstructive pulmonary disease, chronic kidney disease, liver disease, malignancy, asthma, smoking history, and thyroid disease.
In terms of COVID-19 infection severity, we adopted the NIH guidelines for the definitions of asymptomatic, mild, moderate, severe, and critical illness. To meet our inclusion and exclusion criteria, we have excluded those in the “asymptomatic” and “critical illness” category. We have grouped “mild” and “moderate” into “non-severe”, therefore leaving us with two groups: severe and non-severe, and the main difference between these two categories would be the use of oxygen therapy. Finally, we looked at readmission rates within 30 days for patients with PCT.
Study overview and data collection
Designed from a pragmatist viewpoint, this retrospective observational study collected data to respond to the research questions using EPIC (electronic health records), which were deidentified into limited datasets in compliance with both federal and institutional requirements.
All deidentified datasets and statistical findings were stored securely on the study specific SharePoint which is only accessible to approved investigators and the required experts such as the Institution’s Graduate Medical Education Research Team. The dataset was analyzed according to best practices and data types. A summary of findings and statistical processes was stored in the study specific SharePoint, and data validation was carried out by the investigators. We examined data from EPIC chart review between the years 2020 and 2021. Our focus was on patients diagnosed with COVID-19 who were admitted to the inpatient setting and who met our non-exertional tachycardia inclusion and exclusion criteria.
Of the 10,051 patients admitted with a diagnosis of COVID-19, 8,626 patients were excluded specifically for this study, leaving us with 1,425 participants (Fig. 1). A descriptive analysis was performed to analyze the prevalence of tachycardia in post-COVID-19 within various patient demographics and comorbidities. Furthermore, association with the severity of COVID-19 infection and mortality were also incorporated in the statistical study. Finally, a control group was obtained to analyze if there is a significant association of post-COVID-19 and sinus tachycardia.
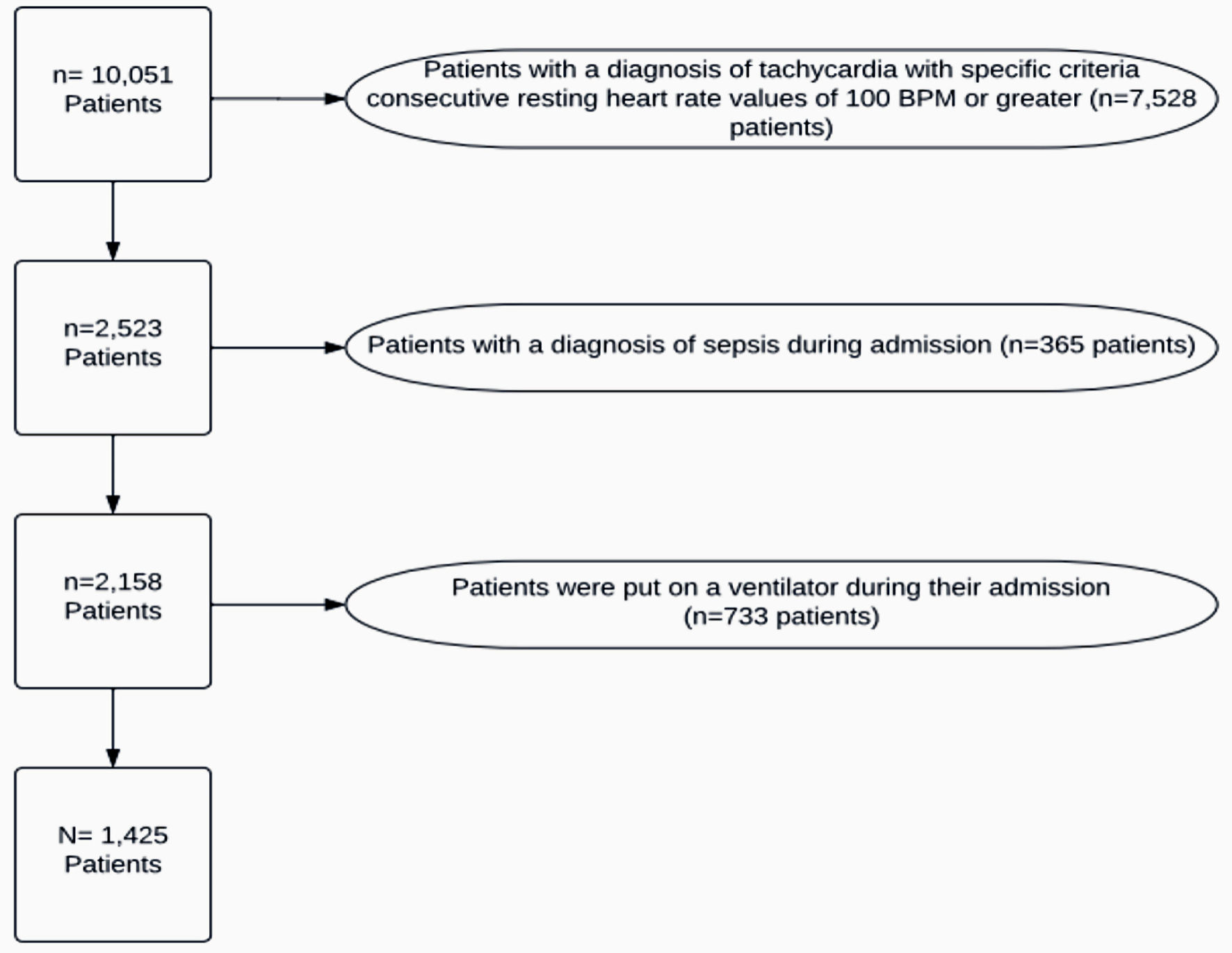 Click for large image | Figure 1. Patient exclusions. BPM: beats per minute. |
Statistical analysis
The primary outcome consisted of the percentage of patients with PCT. Prevalence was calculated within the different points of time relative to the date of discharge. Secondary observational outcomes were analyzed using multinomial logistic regression and cross-tabulation. For each cross-tabulation table, a corresponding Pearson Chi-square analysis was performed. Symmetric measures with Phi and Cramer’s V were also included.
| Results | ▴Top |
A total of 1,425 patients were found to have PCT from September 2018 to March 2022 (Fig. 1). Subjects were in vicinity to Northeast Georgia Medical Center, therefore making Hall County the main location in reference to the subjects’ demographics.
Prevalence of tachycardia
Based on our inclusion and exclusion criteria, sinus tachycardia after resolution of COVID-19 infection was present in 26.3% of patients at day of discharge, 30.8% of patients at 1 day before discharge, 30.3% of patients at 3 days before discharge, 28.8% of patients at 7 days before discharge, and 24.7% of patients at 10 days before discharge (Fig. 2). The formal diagnosis of post-COVID-19 syndrome or long COVID-19 using the International Classification of Diseases, 10th Revision (ICD-10) codes was lacking in terms of documentation, therefore limiting the investigation of sinus tachycardia with this method.
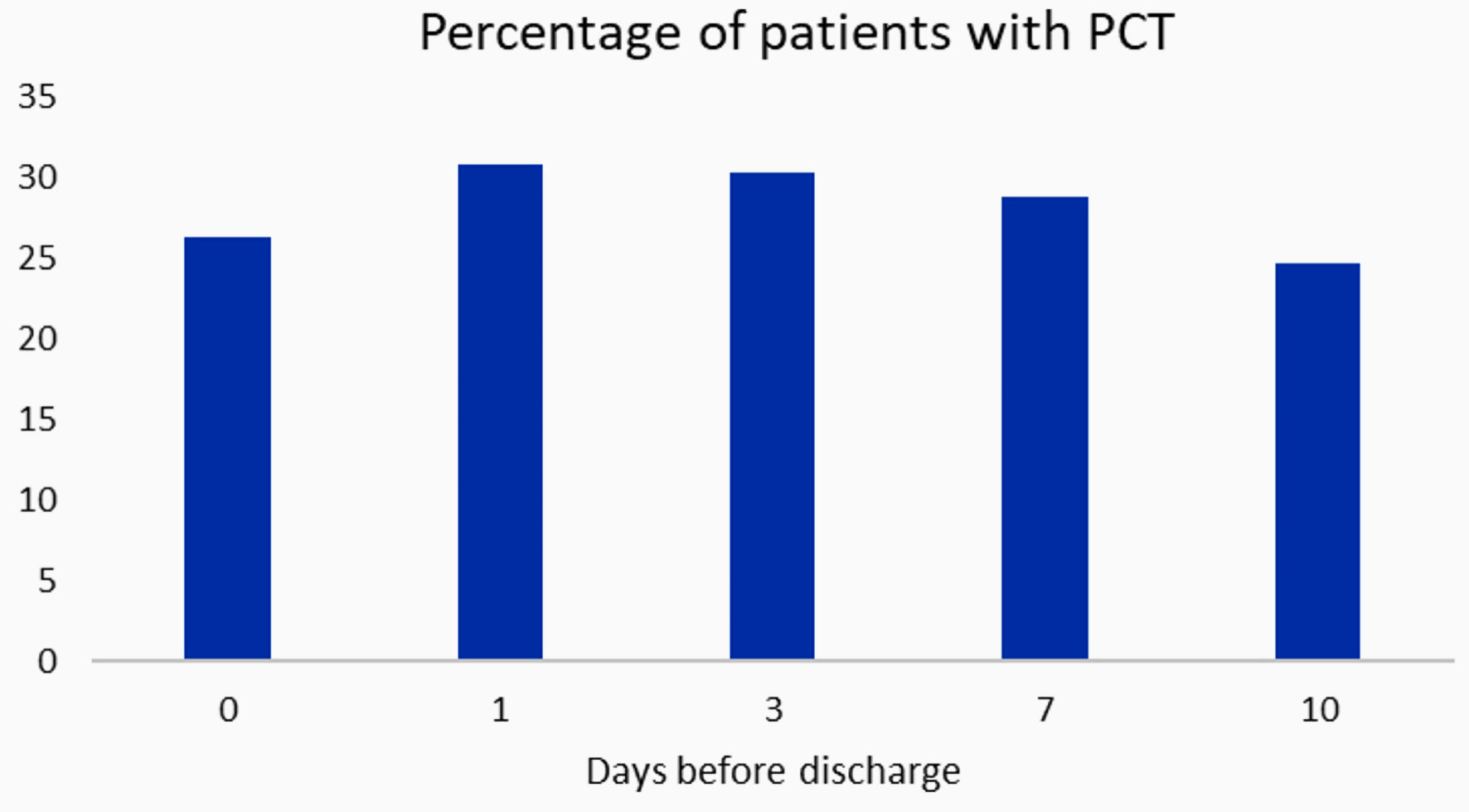 Click for large image | Figure 2. Percentage of patients with post-COVID-19 tachycardia (PCT). COVID-19: coronavirus disease 2019. |
Clinical characteristics
To determine an association between demographics and the propensity of developing PCT, we have looked at age, sex, race, length of stay, tobacco use, financial status and severity of COVID-19 (non-severe or severe). In comparison with “other” type of insurance, patients with commercial type insurance were more likely to develop PCT (Exp(B) = 3.163, P value = 0.001) (Table 2).
 Click to view | Table 2. Association of Demographics and Clinical Characteristics for Developing Post-COVID-19 Tachycardia |
From the regression analysis, patients with commercial insurance had increased chances of developing PCT (Exp(B) = 3.16, P < 0.001) as compared to base (Table 2). “Other” type of insurance had a standardized residual of 3.8 compared to the other groups. The standardized residual for commercial, Medicaid, Medicare and self-pay were 1.4, 1.5, -3.3, and 1.0, respectively (Table 3). In comparison with “never smoked”, patients with “unknown” smoking status were more likely to develop PCT (Exp(B) = 3.297, P value = 0.016). A shorter length of stay was significantly correlated with PCT (Exp(B) = 0.994, P < 0.001).
 Click to view | Table 3. Secondary Outcomes (Race, Financial Status, Age, Gender, Comorbidities, COVID-19 Severity and Readmission Rate) |
Age did not affect PCT (P = 0.131); however, it was found to have a significant association with PCT. The age group of less than 65 had more propensity to experience PCT with a standardized residual of 2.2 compared to -2.5 and -0.9 from age groups 65 - 75 and 75 or more, respectively (Table 3). Race and gender did not have a significant association with PCT (P value = 0.542 and 0.470, respectively) (Table 3).
Comorbidities
When grouping individuals based on number of comorbidities, individuals with fewer comorbidities are most likely to experience PCT (Table 3). Patients with zero comorbidities were more than expected to experience PCT (z = 2.2). However, patients with one and three comorbidities were less than expected to experience PCT (z = 1.5 and z = 1.8) Patients with two and more than four comorbidities were equally expected to experience PCT.
Furthermore, patients with one comorbidity were analyzed to determine which diagnosis was associated with PCT. The purpose of this analysis was also to validate the accuracy of retrospective chart review in terms of misdiagnosis. The most common diagnosis within the “one comorbidity” category was diabetes.
COVID-19 infection severity
There is a significant association between COVID-19 infection severity and tachycardia. Patients with less severe COVID-19 are more likely to experience PCT (Table 3). Patients who had a “less severe” COVID-19 infection had a larger standardized residual value (1.7) compared to “more severe” (-2.0).
Readmission
Patients with PCT had a standardized residual of 2.6 compared to -1.0 from those who did not have PCT (Table 3).
| Discussion | ▴Top |
In this study, the main observation was to determine the potential existence for PCT as a separate phenomenon from other post-COVID-19 related dysautonomia. The number of patients with PCT in our sample of interest was an average of 28.18%. The prevalence was more prominent at 1 day before discharge with the highest percentage (30.8%) as shown in Figure 2. Since patients can remain COVID-19 positive for a long period of time, our general practice does not advocate for repeat tests, and medical providers use clinical judgment instead to discharge patients who are presumed to have achieved infection resolution. With our stringent inclusive and exclusive criteria, the findings strongly suggest that PCT is possibly a separate entity from COVID-19 dysautonomia, which differs from what has been discussed in the literature.
In terms of clinical characteristics, our sample of interest showed a strong association of PCT with patients of age group less than 65 years which coincides with other post-COVID-19 dysautonomia disorders [16]. Additionally, PCT shared a significant association with patients that experienced a shorter length of stay, unknown smoking status, and carrying commercial insurance compared to other demographic characteristics. Less severe COVID-19, defined as unsupported oxygen saturation 90% or greater, and patients experiencing 30-day readmissions were also more likely to experience PCT. A shorter length of stay in the hospital was a finding that could be associated with the increased propensity to PCT in patients with less severe infections. Furthermore, this study also showed that patients with less comorbidities were more commonly associated with PCT. This characteristic of increased association of PCT with young healthier patients that experienced milder infectious courses has been observed previously in literature [8, 13]. We are not able to explain the pathophysiology of why young healthy patients with less severe COVID-19 infection are more likely to develop PCT, but we do theorize that it could be related to the autonomic nervous system (ANS) injury [8, 17]. More severe cases of COVID-19 infections could lead to a larger inflammatory response and severe cytotoxic injury dysregulating the ANS and therefore a lower probability of causing dysautonomia. This concept could be similar to the effects of aging in blunting of ANS [18]. Higher readmission rates coincide with post-COVID-19 syndrome and its lingering symptoms [19].
As mentioned previously, within etiologies of post-COVID-19 dysautonomia, PCT could potentially be a phenomenon separate from POTS and IST. PCT differs from POTS due to elevated heart rates at rest without association with position, and it differs from IST due to asymptomatic persistent tachycardia and having no association to a primary cause [9]. According to Aranyo et al, about 20% of patients with post-COVID-19 syndrome developed concomitant IST [8]. Interestingly, the population of interest were composed of young women, minimal comorbidities, and mild COVID-19 infection which aligns with the clinical characteristics in our study except for gender. Similarly, there has been a rising number of cases thought to be associated with POTS with similar clinical characteristics [16]. There are numbers of studies reinforcing the idea of post-COVID-19 related complications and symptomology, however few of these studies provide comprehensive evidence to the existence of a true cardiovascular component to the post-infection phase [20].
Multiple distinct pathways were theorized to be the candidates for the pathophysiology of cardiovascular involvement post-COVID-19. In the acute phase, with the concept of angiotensin-converting enzyme 2 receptors, COVID-19 infection can affect the heart by the resultant uncontrolled cytokine release, cytotoxic injury, endothelial involvement, thrombo-inflammation, and dysregulation of renin-angiotensin-aldosterone system [5]. Direct myocardial injury and cardiovascular microthrombi were also speculated, but data are not in their favor [21]. Conversely, in the post-acute phase, persistent viral residues in the heart could cause a chronic inflammatory state, which is thought to be intensified by adipokines release in obese patients leading to myocardial fibrosis and its consequences [5].
Indirect pathways that involve the cardiovascular system could be explained by dysautonomia in COVID-19-infected patients. Brainstem dysfunction is one of the causes since it involves the sympathetic outflow [11]. Another major hypothesis is molecular mimicry and its autoimmune response, specifically G-protein-coupled receptors antibodies, which can cause an increased sympathetic tone [5, 20]. The autoimmune mechanism has been theorized to be associated with COVID-19-related POTS as well, and the data could also be an explanation of the higher prevalence in women due to the pathophysiology behind autoimmune diseases. Further studies are necessary to predict a causal relationship between post-COVID-19 syndrome and PCT.
Limitations/strengths
Our study has specific strengths and limitations. One strength involves the stringency and robustness of our inclusion and exclusion criteria to differentiate PCT as much as possible from the overlapping features of both IST and POTS. Another strength is that this is one of the first studies to look at PCT as its own new entity, which can potentially allow other clinicians and researchers to have a better understanding of the complexities of post-COVID-19 complications. In terms of limitations, we did not exclude myocarditis as a common result from COVID-19. The constellation of symptoms associated with myocarditis is rather extensive and could have assimilated PCT in some cases, however diagnosing myocarditis can be tricky especially when almost every patient with COVID-19 infection had elevated troponins, and echocardiograms were not the standard workup. A myocardial biopsy, which is the gold standard diagnostic approach, was not available within the access of this study. Another limitation is that it is based on a retrospective chart review which also limits the accuracy and amount of data collected based on shortcomings associated with charting, and diagnostic documentation.
Conclusions
PCT is likely a separate entity from IST and POTS, and an important entity under the umbrella of post-COVID-19 syndrome. It warrants further studies to elucidate the underlying pathophysiology and to confirm its presence as a distinct entity.
Acknowledgments
We would like to thank the Graduate Medical Education research team and Dr. Louise Jones from Northeast Georgia Medical Center for supporting this research study.
Financial Disclosure
This study did not receive any funding.
Conflict of Interest
The authors declare there is no conflict of interest.
Informed Consent
This is a retrospective chart review that reviewed secondary data. Therefore, the IRB waived the need for informed consent from the participants involved in this study.
Author Contributions
JW was responsible for writing the background and methodology, data collection, and manuscript writing, developing the background and methodology of the study in its origination. DP assisted as the faculty sponsor of the project with reviewing and editing of the entire manuscript and helped with clinical interpretations of data analysis. SR was responsible as the methodologist of the project and assisted in development of all aspects of the manuscript and ethical compliance with IRB submission and review. AR served as the statistician of the study and worked with data cleaning, organization, and analysis. AR also was responsible for data interpretation and assisted with writing the results section of the manuscript. OA assisted the primary author, JW, in all aspects of writing the background and methodology, data collection, and manuscript writing.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Yong SJ. Long COVID or post-COVID-19 syndrome: putative pathophysiology, risk factors, and treatments. Infect Dis (Lond). 2021;53(10):737-754.
doi pubmed pmc - Pavli A, Theodoridou M, Maltezou HC. Post-COVID syndrome: incidence, clinical spectrum, and challenges for primary healthcare professionals. Arch Med Res. 2021;52(6):575-581.
doi pubmed pmc - COVID-19 Rapid Guideline: Managing the long-term effects of COVID-19. National Institute for Health and Care Excellence (NICE). 2020. Accessed January 22, 2024. http://www.ncbi.nlm.nih.gov/books/NBK567261/.
- Raj L, Maidman SD, Adhyaru BB. Inpatient management of acute decompensated heart failure. Postgrad Med J. 2020;96(1131):33-42.
doi pubmed - Raman B, Bluemke DA, Luscher TF, Neubauer S. Long COVID: post-acute sequelae of COVID-19 with a cardiovascular focus. Eur Heart J. 2022;43(11):1157-1172.
doi pubmed pmc - Huang C, Huang L, Wang Y, Li X, Ren L, Gu X, Kang L, et al. 6-month consequences of COVID-19 in patients discharged from hospital: a cohort study. Lancet. 2021;397(10270):220-232.
doi pubmed pmc - Nalbandian A, Sehgal K, Gupta A, Madhavan MV, McGroder C, Stevens JS, Cook JR, et al. Post-acute COVID-19 syndrome. Nat Med. 2021;27(4):601-615.
doi pubmed pmc - Aranyo J, Bazan V, Llados G, Dominguez MJ, Bisbal F, Massanella M, Sarrias A, et al. Inappropriate sinus tachycardia in post-COVID-19 syndrome. Sci Rep. 2022;12(1):298.
doi pubmed pmc - Stahlberg M, Reistam U, Fedorowski A, Villacorta H, Horiuchi Y, Bax J, Pitt B, et al. Post-COVID-19 tachycardia syndrome: a distinct phenotype of post-acute COVID-19 syndrome. Am J Med. 2021;134(12):1451-1456.
doi pubmed pmc - Henning A, Krawiec C. Sinus Tachycardia. In: StatPearls. Treasure Island (FL) ineligible companies, 2024.
pubmed - Goldstein DS. The possible association between COVID-19 and postural tachycardia syndrome. Heart Rhythm. 2021;18(4):508-509.
doi pubmed pmc - Ormiston CK, Swiatkiewicz I, Taub PR. Postural orthostatic tachycardia syndrome as a sequela of COVID-19. Heart Rhythm. 2022;19(11):1880-1889.
doi pubmed pmc - Novak P, Giannetti MP, Weller E, Hamilton MJ, Mukerji SS, Alabsi HS, Systrom D, et al. Network autonomic analysis of post-acute sequelae of COVID-19 and postural tachycardia syndrome. Neurol Sci. 2022;43(12):6627-6638.
doi pubmed pmc - Gopinathannair R, Olshansky B. Management of tachycardia. F1000Prime Rep. 2015;7:60.
doi pubmed pmc - CDC. Coronavirus Disease 2019 (COVID-19). Centers for Disease Control and Prevention. 2020. Accessed February 5, 2024. https://www.cdc.gov/coronavirus/2019-ncov/long-term-effects/post-covid-appointment/index.html.
- Davis HE, Assaf GS, McCorkell L, Wei H, Low RJ, Re'em Y, Redfield S, et al. Characterizing long COVID in an international cohort: 7 months of symptoms and their impact. EClinicalMedicine. 2021;38:101019.
doi pubmed pmc - Petersen MS, Kristiansen MF, Hanusson KD, Danielsen ME, B AS, Gaini S, Strom M, et al. Long COVID in the faroe islands: a longitudinal study among nonhospitalized patients. Clin Infect Dis. 2021;73(11):e4058-e4063.
doi pubmed pmc - Cheshire WP, Freeman R, Gibbons CH, Cortelli P, Wenning GK, Hilz MJ, Spies JM, et al. Electrodiagnostic assessment of the autonomic nervous system: A consensus statement endorsed by the American Autonomic Society, American Academy of Neurology, and the International Federation of Clinical Neurophysiology. Clin Neurophysiol. 2021;132(2):666-682.
doi pubmed - Sotoodeh Ghorbani S, Taherpour N, Bayat S, Ghajari H, Mohseni P, Hashemi Nazari SS. Epidemiologic characteristics of cases with reinfection, recurrence, and hospital readmission due to COVID-19: A systematic review and meta-analysis. J Med Virol. 2022;94(1):44-53.
doi pubmed pmc - Bisaccia G, Ricci F, Recce V, Serio A, Iannetti G, Chahal AA, Stahlberg M, et al. Post-acute sequelae of COVID-19 and cardiovascular autonomic dysfunction: what do we know? J Cardiovasc Dev Dis. 2021;8(11):156.
doi pubmed pmc - Bois MC, Boire NA, Layman AJ, Aubry MC, Alexander MP, Roden AC, Hagen CE, et al. COVID-19-associated nonocclusive fibrin microthrombi in the heart. Circulation. 2021;143(3):230-243.
doi pubmed pmc
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


