| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website http://www.cardiologyres.org |
Case Report
Volume 8, Number 2, April 2017, pages 77-80
Transient Left Bundle Branch Block due to Severe Hyperkalemia
Kishore Kumara, b, Madhavi Biyyama, Amandeep Singha, Bharat Bajantria, Hassan Tariqa, Suresh Kumar Nayudua, Sridhar Chilimuria
aDepartment of Medicine, Bronx Lebanon Hospital Center, Bronx, NY 10457, USA
bCorresponding Author: Kishore Kumar, Department of Medicine, Bronx Lebanon Hospital Center, Bronx, NY 10457, USA
Manuscript accepted for publication March 21, 2017
Short title: Transient Left Bundle Branch Block
doi: https://doi.org/10.14740/cr538w
| Abstract | ▴Top |
Hyperkalemia is a potentially life-threatening electrolyte imbalance that can lead to sudden death from cardiac arrhythmias and asystole. We present a case of transient left bundle branch block pattern on an electrocardiogram (ECG) secondary to hyperkalemia in a patient with history of end-stage renal disease. A 52-year-old man presented to the emergency room (ER) with chief complaints of weakness and lethargy after missing his regularly scheduled session of hemodialysis. A 12-lead ECG in the ER showed sinus tachycardia at 118 beats/min, wide QRS complexes, peaked T waves and left bundle branch block-like pattern. The initial basic metabolic panel revealed a serum potassium level of 8.8 mEq/L. Subsequently, the patient underwent emergent hemodialysis. Serum chemistry after hemodialysis showed improvement in serum potassium to 4.3 mEq/L. Repeat ECG performed after correcting potassium showed dissolution of left bundle branch block finding.
Keywords: Left bundle branch block; Hyperkalemia; ECG
| Introduction | ▴Top |
Hyperkalemia is a medical emergency which may lead to sudden cardiac death secondary to cardiac arrhythmias if left untreated. Any patient with history and physical examination which raise clinical suspicion of hyperkalemia requires an immediate 12-lead electrocardiogram (ECG) to ascertain whether ECG manifestations of hyperkalemia are present or not. Hyperkalemia is considered to be severe when the serum potassium level is 7.0 mEq/L or greater. The mortality among patients with severe hyperkalemia can be as high as 67% if not corrected in a timely manner [1, 2].
| Case Report | ▴Top |
A 52-year-old man presented to the emergency room with complaints of weakness and lethargy. According to the patient, the symptoms started after he skipped a scheduled hemodialysis session for unknown reason. His medical history included end-stage renal disease requiring hemodialysis three times/week, diabetes mellitus type 2, hypertension, congestive heart failure, chronic obstructive pulmonary disease, acquired immunodeficiency syndrome (AIDS) and chronic hepatitis C. He is an active smoker and enrolled in methadone maintenance program in view of prior intravenous drug use. His home medications included aspirin, atorvastatin, lisinopril, hydralazine, metoprolol, isosorbide mononitrate, insulin, albuterol, fluticasone/salmeterol and tiotropium. On arrival to the emergency room, his vitals were heart rate of 118 beats/min regular rhythm, blood pressure of 110/50 mm Hg, respiratory rate of 16 breaths/min and an oxygen saturation of 87% while breathing on 2 L/min of nasal oxygen. The cardiopulmonary examination showed bilateral air entry without any adventitious sounds and normal heart sounds without murmurs, gallop or rub. The abdomen was soft with no organomegaly and normal bowel sounds. Extremities were warm and well perfused without edema, cyanosis, or clubbing. Neurologic examination was unremarkable. A 12-lead ECG (Fig. 1) showed the hallmark features of hyperkalemia including left bundle branch block, hyper-acute T waves, sinus tachycardia at 118 beats/min and wide QRS complex. The initial laboratory investigations showed serum sodium of 123 mEq/L, potassium of 8.8 mEq/L, blood urea nitrogen of 133 mg/dL, creatinine of 10.6 mg/dL, pH 6.9, pCO2 of 56 mm Hg, pO2 of 75 mm Hg and HCO3- of 10.7 mmol/L. He was given a cocktail of intravenous calcium gluconate, insulin, dextrose, and bicarbonate for severe hyperkalemia; however, his repeat serum potassium level remained elevated for which he underwent emergent hemodialysis. A repeat ECG (Fig. 2) after hemodialysis showed a resolution of the left bundle branch pattern along with normal rhythm and axis, left ventricular hypertrophy with strain pattern and persistent prolonged QT interval. A repeat serum chemistry after hemodialysis showed improvement in serum potassium level to 4.3 mEq/L. The persistent prolongation of the QT interval was attributed to chronic use of methadone.
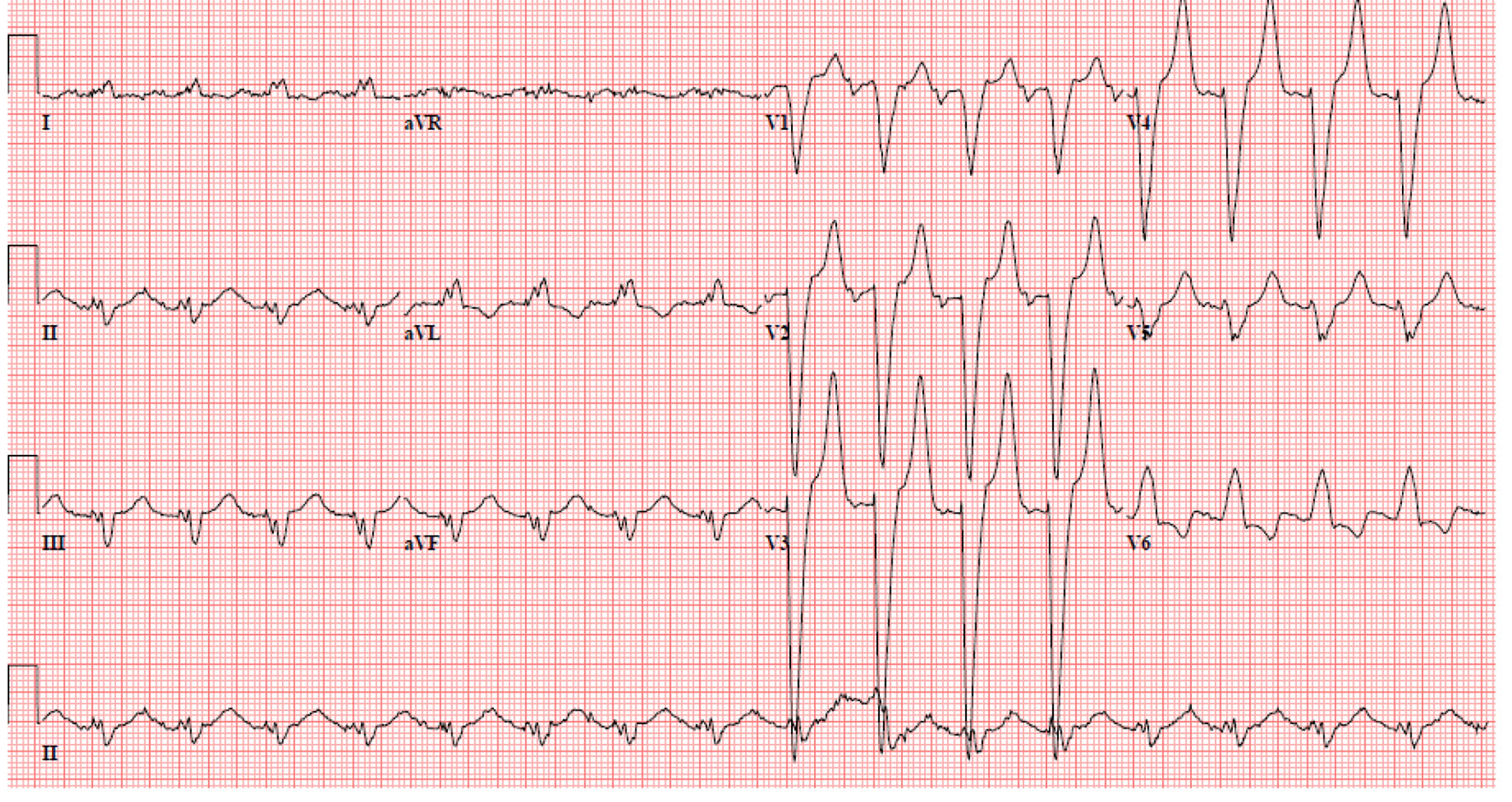 Click for large image | Figure 1. Electrocardiogram showed left bundle branch pattern, along with tall T waves and wide QRS complex. |
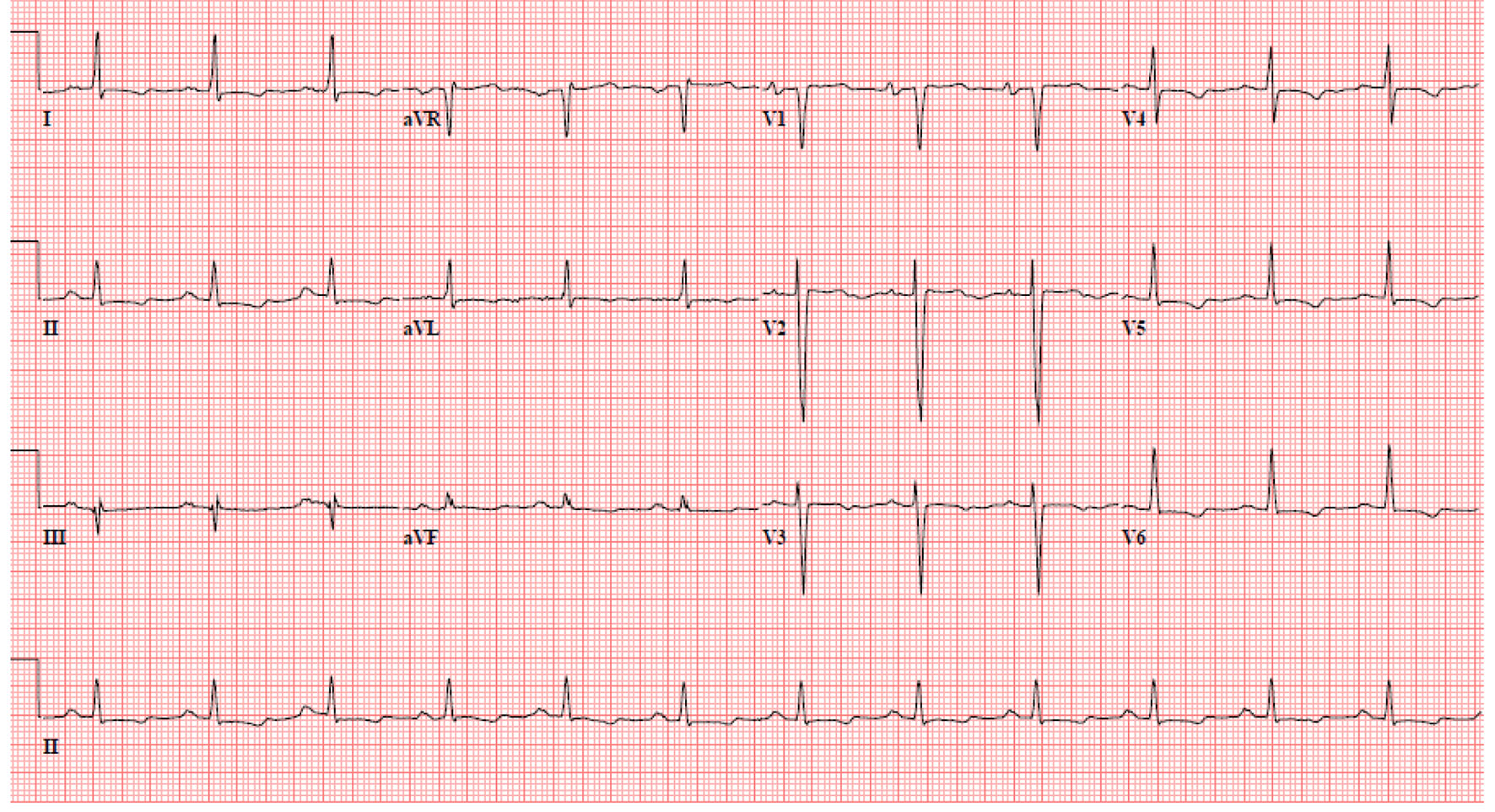 Click for large image | Figure 2. Electrocardiogram after the correction of hyperkalemia. |
| Discussion | ▴Top |
Hyperkalemia is one of the most common causes of the cardiac arrhythmias seen in clinical practice. It is important to note that the relationship between potassium levels and ECG findings varies between patients and many less known and less recognized ECG changes associated with hyperkalemia are reported in the literature which includes hemiblock, right bundle branch block, left bundle branch block, bifascicular block, or trifascicular block due to depressed intraventricular conduction. Although the left bundle branch block due to severe hyperkalemia has been reported previously but its exact prevalence and relation to potassium level is not known. Geriatric population in particular is vulnerable to develop severe hyperkalemia due to multiple factors, of which polypharmacy is one of the most common contributing factors. Beta-adrenergic blocker, NSAID, aldosterone antagonist, angiotensin-converting enzyme inhibitor, angiotensin II receptor blocker and digoxin are common medications known to contribute to hyperkalemia. Comorbid conditions [3, 4] like diabetes mellitus, hypertension, coronary artery disease and chronic kidney disease often further impede homeostatic function which magnifies the risk of developing hyperkalemia. The ratio of intracellular to extracellular potassium concentration is the primary determinant of cell membrane potential in most cells [5]. Normally, intracellular concentration is approximately 120 mEq/L, whereas extracellular potassium concentration is approximately 4 mEq/L. The contribution of potassium to resting membrane potential is related to this ratio of intracellular to extracellular potassium; thus, small changes in extracellular potassium can result in large changes in the intracellular to extracellular potassium ratio, hence result in large changes in resting membrane potential. The resting membrane potential is important in all electrically active cells, including neurons, voluntary and involuntary muscles. Total body potassium levels are regulated mostly by the kidneys, with only 5-10% of ingested potassium excreted in the feces. The renal excretion of potassium is determined by the rate of potassium filtration across the glomerular basement membrane and by the rate of its secretion and resorption in the distal tubules of the nephron. This equilibrium is regulated by insulin [6-8], catecholamines [9, 10] and to a lesser extent, by acid-base balance [11-13] and plasma tonicity.
The sequence of ECG manifestations is directly linked to severity of hyperkalemia as depicted in Table 1 [14]. The earliest ECG manifestation of hyperkalemia is the appearance of narrow-based, peaked T waves. These T waves are of relatively short duration, approximately 150 - 250 ms. Peaked T waves are usually seen at potassium concentrations greater than 5.5 mEq/L and are best seen in leads II, III, and V2 through V4, but are present in only 22% of patients with hyperkalemia. Hyperkalemia also causes delayed intraventricular and atrioventricular conduction and as the intraventricular conduction delay worsens, the QRS complex may take on the appearance of a left or right bundle branch block configuration. A clue that these ECG changes are due to hyperkalemia, and not to bundle branch disease, is that, in hyperkalemia the conduction delay persists throughout the QRS complex and not just in the initial or terminal portions, as seen at left and right bundle branch block. Another rare manifestation of hyperkalemia is ST segment elevation or pseudo infarction [15-17]. For the management of hyperkalemia most authorities recommend immediate treatment when ECG changes are present or when serum potassium levels are greater than 6.5 mEq/L regardless of the ECG findings. The treatment for hyperkalemia can be divided into three distinct categories: firstly, antagonize the effects of hyperkalemia at the cellular level (membrane stabilization); secondly, decrease serum potassium levels by promoting the influx of potassium into cells throughout the body; and lastly, remove potassium from the body through kayexalate [18, 19] or hemodialysis. The effects of intravenous calcium therapy occur within 1 - 3 min and last for only 30 - 60 min [20]. Therefore, more definitive treatment is needed to lower serum potassium levels. Calcium gluconate is the preferred preparation of intravenous calcium. The recommended dose of 10 mL of a 10% calcium gluconate solution infused over 2 - 3 min antagonizes myocardial effects of hyperkalemia [21, 22]. If all pharmacological management fails, then the next step would be emergent dialysis. Hyperkalemia is often a reason of elimination of useful medications such as aldosterone antagonist, angiotensin-converting enzyme inhibitor, and angiotensin II receptor blocker, which has a mortality benefit in some chronic conditions like congestive heart failure and chronic kidney disease. The emergence of new potassium binders allows the continued use of medications such as renin-angiotensin aldosterone system inhibitors even in patients who are prone to develop hyperkalemia. This may result in improved outcomes in patients with cardiovascular and renal diseases. Lastly, thorough knowledge of the ECG manifestations of hyperkalemia is imperative to ensure prompt treatment of this potentially life-threatening condition.
 Click to view | Table 1. Electrocardiographic Manifestations of Serum Hyperkalemia Relative to Serum Potassium Level [14] |
| References | ▴Top |
- Weisberg LS. Management of severe hyperkalemia. Crit Care Med. 2008;36(12):3246-3251.
doi pubmed - Perazella MA, Mahnensmith RL. Hyperkalemia in the elderly: drugs exacerbate impaired potassium homeostasis. J Gen Intern Med. 1997;12(10):646-656.
doi - Parham WA, Mehdirad AA, Biermann KM, Fredman CS. Hyperkalemia revisited. Tex Heart Inst J. 2006;33(1):40-47.
pubmed - Michelis MF. Hyperkalemia in the elderly. Am J Kidney Dis. 1990;16(4):296-299.
doi - Guyton AC, Hall JE. Textbook of medical physiology, 9th ed. Philadelphia: WB Saunders; 1996. p. 375-380.
- Ahmed J, Weisberg LS. Hyperkalemia in dialysis patients. Semin Dial. 2001;14(5):348-356.
doi pubmed - Clausen T, Everts ME. Regulation of the Na,K-pump in skeletal muscle. Kidney Int. 1989;35(1):1-13.
doi pubmed - DeFronzo RA, Felig P, Ferrannini E, Wahren J. Effect of graded doses of insulin on splanchnic and peripheral potassium metabolism in man. Am J Physiol. 1980;238(5):E421-427.
pubmed - DeFronzo RA, Bia M, Birkhead G. Epinephrine and potassium homeostasis. Kidney Int. 1981;20(1):83-91.
doi pubmed - Rosa RM, Silva P, Young JB, Landsberg L, Brown RS, Rowe JW, Epstein FH. Adrenergic modulation of extrarenal potassium disposal. N Engl J Med. 1980;302(8):431-434.
doi pubmed - Adrogue HJ, Madias NE. Changes in plasma potassium concentration during acute acid-base disturbances. Am J Med. 1981;71(3):456-467.
doi - Magner PO, Robinson L, Halperin RM, Zettle R, Halperin ML. The plasma potassium concentration in metabolic acidosis: a re-evaluation. Am J Kidney Dis. 1988;11(3):220-224.
doi - Perez GO, Oster JR, Vaamonde CA. Serum potassium concentration in acidemic states. Nephron. 1981;27(4-5):233-243.
doi pubmed - Mattu A, Brady WJ, Robinson DA. Electrocardiographic manifestations of hyperkalemia. Am J Emerg Med. 2000;18(6):721-729.
doi pubmed - Levine HD, Wanzer SH, Merrill JP. Dialyzable currents of injury in potassium intoxication resembling acute myocardial infarction or pericarditis. Circulation. 1956;13(1):29-36.
doi pubmed - Sweterlitsch EM, Murphy GW. Acute electrocardiographic pseudoinfarction pattern in the setting of diabetic ketoacidosis and severe hyperkalemia. Am Heart J. 1996;132(5):1086-1089.
doi - Ziakas A, Basagiannis C, Stiliadis I. Pseudoinfarction pattern in a patient with hyperkalemia, diabetic ketoacidosis and normal coronary vessels: a case report. J Med Case Rep. 2010;4:115.
doi pubmed - Frohnert PP, Johnson WJ, Mueller GJ, Tauxe WN, McCall JT. Resin treatment of hyperkalemia. II. Clinical experience with a cation exchange resin (calcium cycle). J Lab Clin Med. 1968;71(5):840-846.
pubmed - Frohnert PP, Johnson WJ, Mueller GJ, Tauxe WN, McCall JT. Resin treatment of hyperkalemia. I. Exchange properties of a cation exchange resin (calcium cycle). J Lab Clin Med. 1968;71(5):834-839.
pubmed - Weisberg LS. Potassium homeostasis. In: Carlson RW, Geheb MA (Eds). Principles and Practice of Medical Intensive Care. Philadelphia: Saunders; 1993.
- Bisogno JL, Langley A, Von Dreele MM. Effect of calcium to reverse the electrocardiographic effects of hyperkalemia in the isolated rat heart: a prospective, dose-response study. Crit Care Med. 1994;22(4):697-704.
pubmed - Chamberlain MJ. Emergency treatment of hyperkalemia. Lancet. 1964;18:464-467.
doi
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


