| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website http://www.cardiologyres.org |
Original Article
Volume 10, Number 3, June 2019, pages 172-180
Relationships Between Skin Autofluorescence and Cardio-Ankle Vascular Index in Japanese Male Patients With Metabolic Syndrome
Takashi Hitsumoto
Hitsumoto Medical Clinic, 2-7-7, Takezakicyou, Shimonoseki City, Yamaguchi 750-0025, Japan
Manuscript submitted April 27, 2019, accepted May 30, 2019
Short title: Skin AF and CAVI in Mets
doi: https://doi.org/10.14740/cr878
| Abstract | ▴Top |
Background: An autofluorescence (AF) reader can be used to noninvasively measure tissues that accumulated advanced glycation end-products to diagnose skin AF. This study aimed to clarify the clinical significance of skin AF as a risk factor for cardiovascular disease in Japanese male patients with metabolic syndrome using the cardio-ankle vascular index (CAVI) as a marker of arterial function.
Methods: This cross-sectional study enrolled 261 Japanese male patients with metabolic syndrome without history of cardiovascular disease (mean age, 58 ± 7 years (mean ± standard deviation)). Associations between skin AF and various clinical parameters including CAVI were examined.
Results: Skin AF was significantly positively correlated with CAVI (r = 0.40, P < 0.001). Furthermore, multiple regression analyses revealed that skin AF (β = 0.18, P = 0.002) was selected as an independent subordinate factor for CAVI. Meanwhile, homeostatic model assessment of insulin resistance (HOMA-IR) as a marker of insulin resistance, smoking habits and high-sensitivity C-reactive protein as an inflammation marker were independent variables for either CAVI or skin AF as a subordinate factor. According to the receiver-operating characteristic curve analysis and results of previous reports that determined CAVI of ≥ 9.0 as a diagnostic criterion for vascular failure, skin AF of > 2.7 arbitrary unit is the optimal cut-off point for discriminating high CAVI (area under the curve = 0.718, P < 0.001).
Conclusion: Findings in this study indicate that skin AF may be an important risk factor of cardiovascular disease in Japanese male patients with metabolic syndrome. In addition, the risk value of skin AF was considered as higher than 2.7 arbitrary unit. Further investigations in a large number of prospective studies, including intervention therapies, are required to validate the results in this study.
Keywords: Skin autofluorescence; Cardio-ankle vascular index; Metabolic syndrome; Insulin resistance; Smoking; Inflammation; Male gender; Japanese
| Introduction | ▴Top |
Recent clinical and epidemiological studies indicated that patients with metabolic syndrome (Mets) increased worldwide because of changing lifestyle such as the eating or lack of exercise habits [1, 2]. In addition, the increase of patients with Mets was reportedly associated with increased incidence of cardiovascular disease (CVD), such as ischemic heart disease and stroke [3-5]. Therefore, effective methods should be considered to decrease the number of patients with Mets in daily practice and also explore a novel target factor to prevent CVD events in patients with Mets.
The cardio-ankle vascular index (CAVI) is a novel physiological marker of arteriosclerosis, reflecting arterial stiffness in the aorta, femoral and tibial arteries [6]. In addition, several clinical reports indicate that CAVI reflects endothelial dysfunction considered as reflecting early stage of atherosclerosis [7, 8]. In recent years, a number of clinical data were accumulated regarding the usefulness of CAVI as a CVD risk factor in various types of population including those with Mets [9-12].
Advanced glycation end-products (AGEs) play an important role in the pathophysiology of various diseases. Among the methods used to evaluate AGEs, skin autofluorescence (AF) is known to be a simple and reliable marker in vivo, and recent clinical studies have indicated that skin AF levels are significantly associated with CVD incidence [13-15]. In addition, researchers reported that skin AF levels were significantly higher in patients with Mets than those with Mets [16]. However, data regarding the usefulness of skin AF as a CVD risk factor in patients with Mets are limited. Therefore, this cross-sectional study attempts to clarify the clinical significance of skin AF as a risk factor for CVD in patients with Mets using CAVI.
| Materials and Methods | ▴Top |
Patients
Patients in this study were enrolled between September 2015 and August 2018. The study population comprised 261 Japanese male patients with Mets, who visited the Hitsumoto Medical Clinic. Their mean age was 58 ± 7 years (mean ± standard deviation (SD)). Diagnosis of Mets was evaluated according to the Japanese criteria. Patients with CVD history or those using medications such as antihyperlipidemic, antihypertensive, or antidiabetic drugs were excluded. This study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as the Helsinki Declaration. All patients provided informed consent, and the study protocol was approved by the Institutional Review Board of the Hitsumoto Medical Clinic (approval number: 2015-08).
Diagnosis of Mets
Mets was diagnosed based on the Japanese Committee for the Diagnostic Criteria of Metabolic Syndrome [17]. In the Japanese criteria, Mets was defined as the presence of two or more clinical abnormalities (three abnormalities: dyslipidemia (serum triglyceride concentrations of ≥ 150 mg/dL and/or serum high-density lipoprotein cholesterol concentrations of < 40 mg/dL), elevated blood pressure levels (systolic blood pressure of ≥ 130 mm Hg and/or diastolic blood pressure of ≥ 85 mm Hg) and elevated blood glucose levels (fasting blood glucose of ≥ 110 mg/dL)), in addition to visceral fat obesity (waist circumference of ≥ 85 cm in men and ≥ 90 cm in women). Previous studies indicated that Mets evaluated according to the Japanese diagnosis criteria significantly occurs with coronary atherosclerosis or CVD [3, 18].
Skin AF measurement
Figure 1 shows measuring method of skin AF. Skin AF was automatically measured using a commercial instrument (AGE Reader™; DiagnOptics, Groningen, the Netherlands), as previously described [19, 20]. With patients seated, all measurements were taken at the volar side of the lower arm, approximately 10 - 15 cm below the elbow. AF was defined as the average light intensity per nanometer between 300 and 420 nm. The skin AF levels were expressed in arbitrary unit (AU). The levels of pentosidine, a major AGE component, were measured using skin biopsy at the volar side of the lower arm and appeared to be significantly correlated with skin AF [21]. The validity and reliability of skin AF levels in the Japanese population measured using this method have been previously established [20].
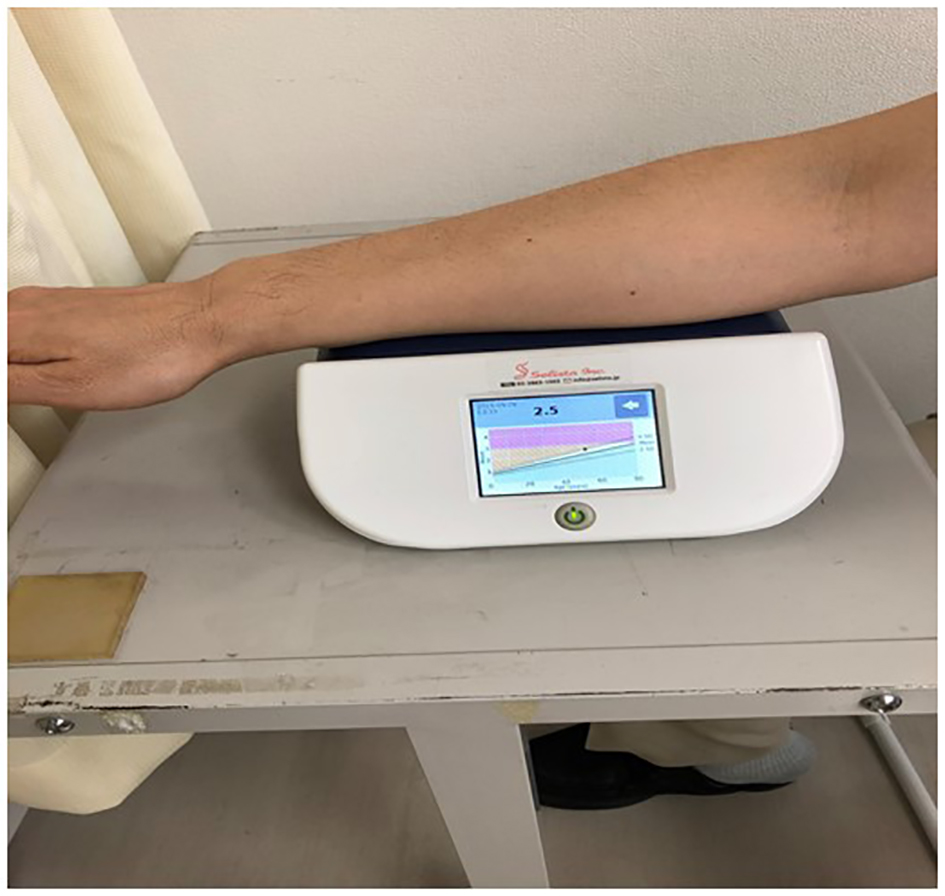 Click for large image | Figure 1. Measuring method of skin AF. Skin AF was automatically measured using a commercial instrument. With patients seated, all measurements were taken at the volar side of the lower arm, approximately 10 - 15 cm below the elbow. AF was defined as the average light intensity per nanometer between 300 and 420 nm. The skin AF levels were expressed in arbitrary unit. AF: autofluorescence. |
CAVI measurement
Figure 2 shows measuring method of CAVI. CAVI was measured using a VaSera CAVI instrument (Fukuda Denshi, Tokyo, Japan) following the previously described methods [6]. Briefly, the brachial and ankle pulse waves were determined using inflatable cuffs with the pressure maintained between 30 and 50 mm Hg to ensure its minimal effect on the systemic hemodynamics. Systemic blood and pulse pressures were simultaneously determined, with the participant in a supine position. CAVI was measured after the participants had rested for 10 min in a quiet room. For statistical evaluation of the CAVI, mean values of the left and right sides were used. The average coefficient of CAVI variation has been shown to be < 5%, which is small enough for clinical use and indicates that CAVI measurement has good reproducibility [22].
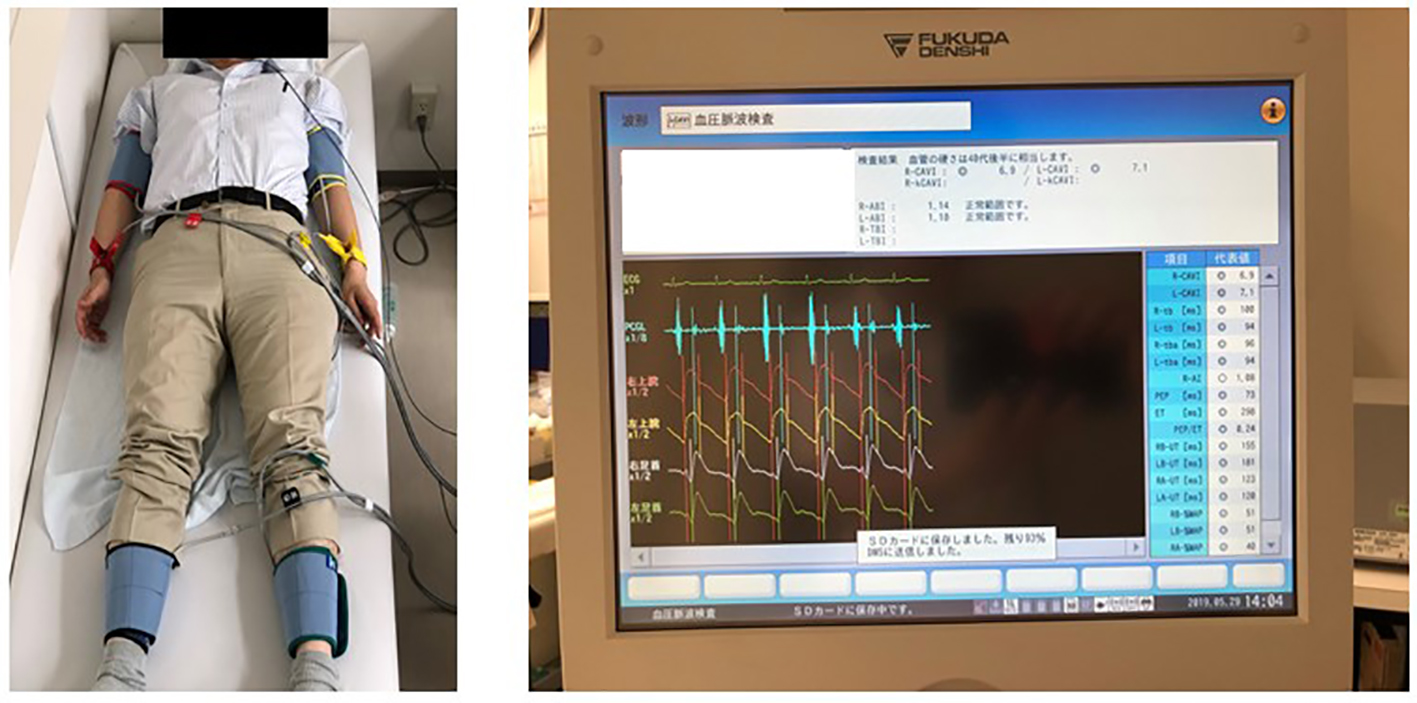 Click for large image | Figure 2. Measuring method of CAVI. The brachial and ankle pulse waves were determined using inflatable cuffs with the pressure maintained between 30 and 50 mm Hg to ensure that the cuff pressure had a minimal effect on the systemic hemodynamics. Blood and pulse pressures were determined simultaneously with the subjects lying supine, after they had first rested for 10 min in a quiet room. CAVI was calculated using the following formula: CAVI = a{(2ρ/ΔP) × ln (Ps/Pd)PWV2} + b, where a and b are constants, ρ is blood density and ΔP is Ps - Pd. CAVI: cardio-ankle vascular index; Ps: systolic blood pressure; Pd: diastolic blood pressure; PWV: pulse wave velocity. |
Estimation of cardiovascular risk factors
The degree of obesity was evaluated using body mass index, calculated as weight (kg) divided by the squared height (m2). Smoker was defined as smoking at least one cigarette per day during the last 1 year, and Brinkmen index (number of smoking (/day) × smoking duration (years)) was used as a marker of smoking exposure. The right brachial blood pressure was measured twice using a mercury sphygmomanometer, with participants in sitting position. An average of two readings was used to determine the systolic and diastolic blood pressures. Blood samples were collected from the antecubital vein in the morning after 12 h of fasting. Total cholesterol and triglyceride levels were measured using standard enzymatic methods. Serum high-density lipoprotein cholesterol levels were measured using selective inhibition. Serum low-density lipoprotein cholesterol levels were calculated using the Friedewald equation [23]. Glucose and insulin levels were measured using the glucose oxidase method and an enzyme immunoassay, respectively. To measure insulin resistance, homeostatic model assessment of insulin resistance (HOMA-IR) was calculated using the following equation [24]: HOMA-IR = fasting glucose concentration (mg/dL) × fasting insulin concentration (µg/mL)/405. High-sensitivity C-reactive protein (hs-CRP) concentration as a marker of inflammation was measured using high-sensitivity, latex-enhanced immunonephelometrics.
Statistical analysis
Data were analyzed using the Stat View-J 5.0 (HULINKS, Tokyo, Japan) and MedCalc for Windows version 14.8.1 (MedCalc Software, Ostend, Belgium) and are expressed as mean ± SD. Between-group comparisons were performed using the Student’s t-test, and the correlation coefficient was estimated using the Pearson or Spearman rank-order correlation analysis. Multivariate analysis was performed using multiple regression analyses. The receiver-operating characteristic curves were constructed, and the maximum Youden index [25] was used to determine the optimal skin AF cut-off levels of high CAVI. A P-value of < 0.05 was considered statistically significant.
| Results | ▴Top |
Study population
Table 1 summarizes patient characteristics of this study. The mean value of skin AF was 2.4 ± 0.6 AU (range, 1.5 - 4.6), and the mean value of CAVI was 8.3 ± 1.3 (range, 6.2 - 12.3). Skin AF and CAVI have nearly normal distribution.
 Click to view | Table 1. Patient Characteristics |
Correlation between factors
The correlations between skin AF and CAVI are shown in Figure 3. Significantly positive correlations were observed between these two clinical parameters. Figure 4 shows the relationships between smoking habits and CAVI or skin AF. Both CAVI and skin AF were significantly higher in smokers than in non-smokers. In addition, both CAVI and skin AF were significantly positively correlated with the Brinkman index. Table 2 presents relationships among CAVI, skin AF and various clinical parameters. Age, systolic blood pressure, fasting blood glucose levels, HOMA-IR, hs-CRP and the number of Mets components were significantly positively correlated with CAVI. On the contrary, age, HOMA-IR and hs-CRP were significantly positively correlated with skin AF.
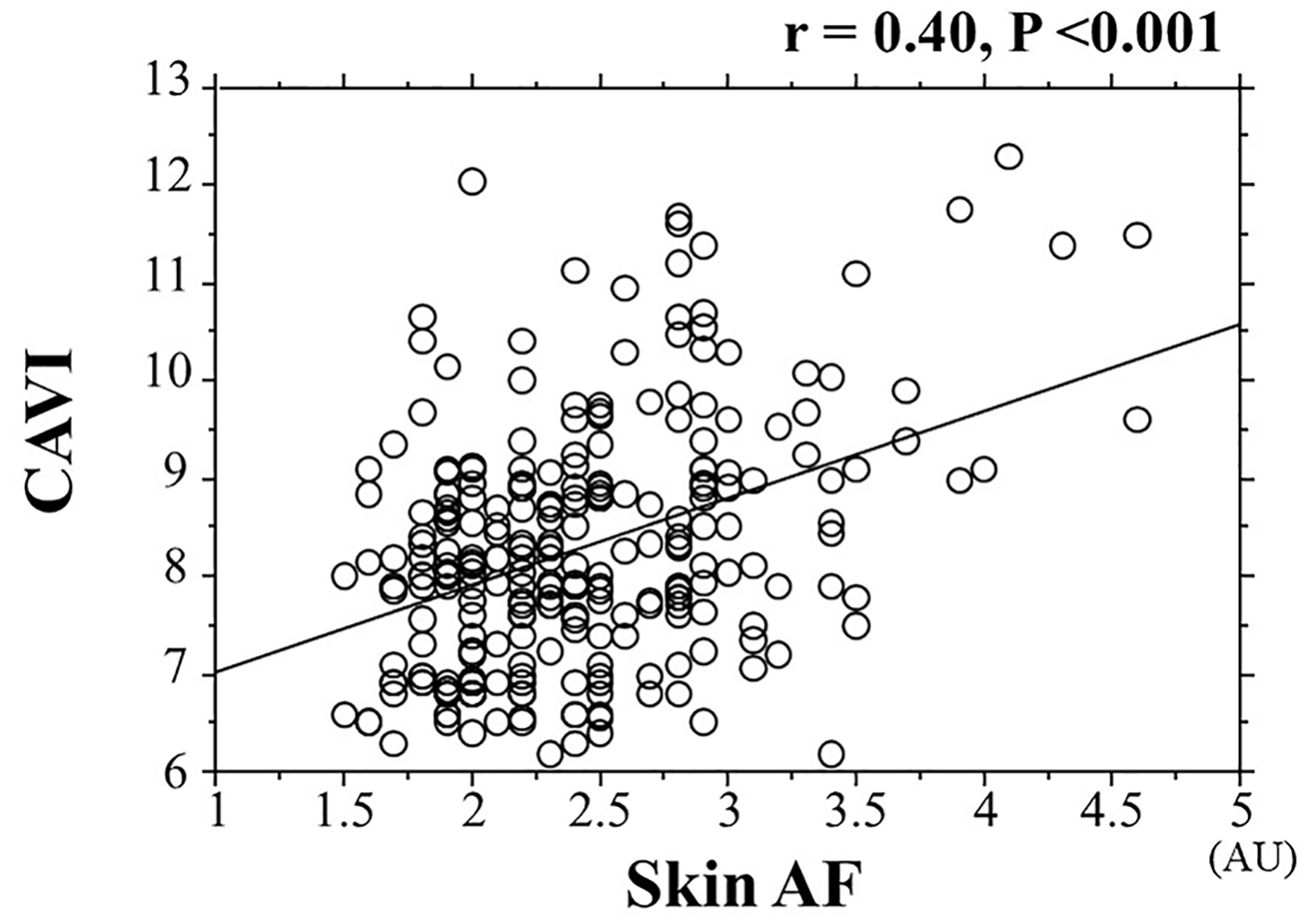 Click for large image | Figure 3. Correlation between skin AF and CAVI. AF: autofluorescence; CAVI: cardio-ankle vascular index; AU: arbitrary unit. |
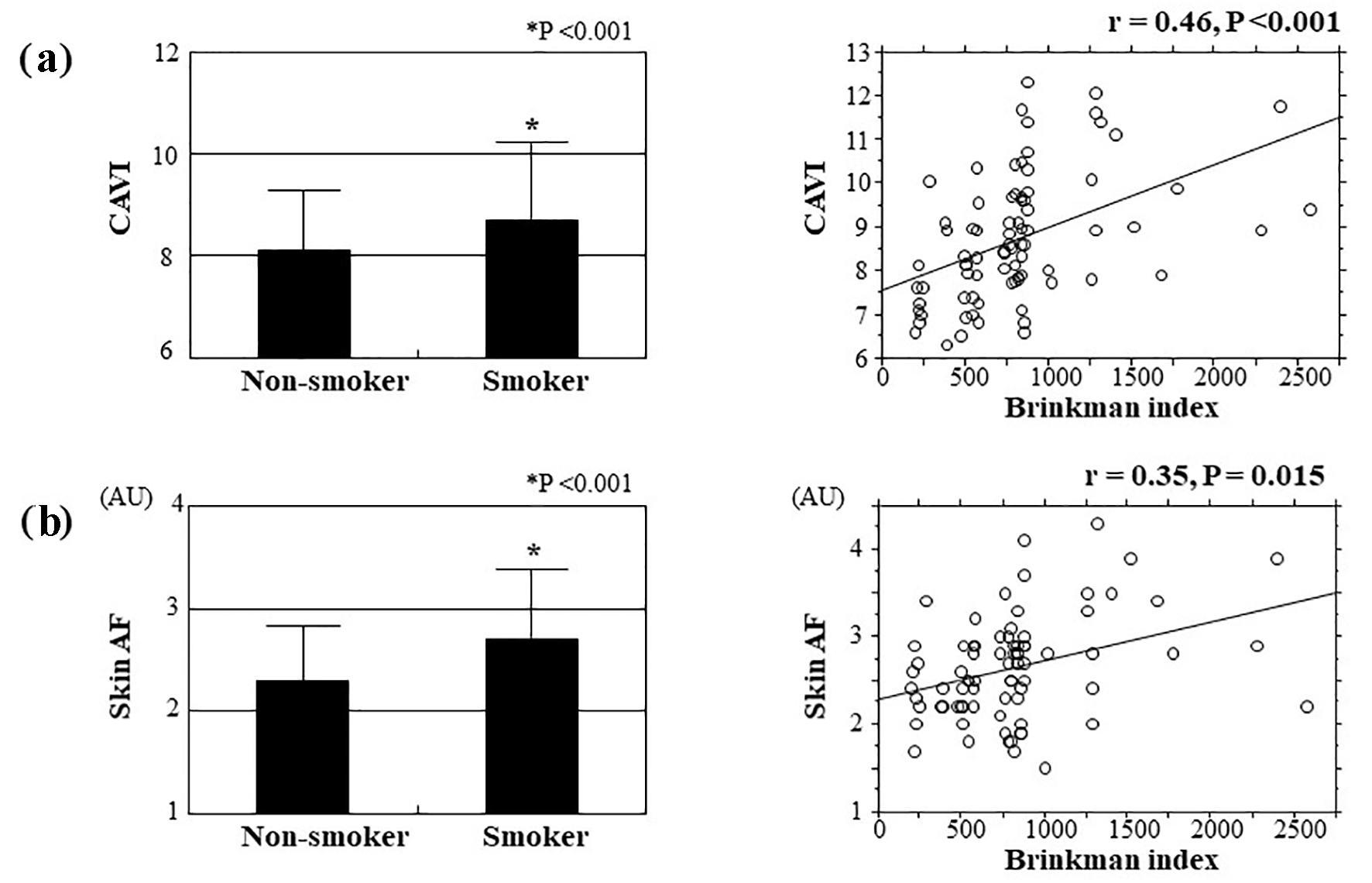 Click for large image | Figure 4. Relations between smoking habits and CAVI and skin AF. Both CAVI (smoker vs. non-smoker: 8.7 ± 1.4 vs. 8.1 ± 1.1, respectively, P < 0.001) and skin AF (smoker vs. non-smoker: 2.7 ± 0.6 AU vs. 2.3 ± 0.5 AU, respectively, P < 0.001) were significantly higher in patients who are smokers than in those who are non-smokers. In addition, both CAVI and skin AF were significantly positively correlated with Brinkman index as a marker of smoking exposure. (a) Relations between smoking habits and CAVI. (b) Relations between smoking habits and skin AF. CAVI: cardio-ankle vascular index; AF: autofluorescence; AU: arbitrary unit. |
 Click to view | Table 2. Relationships Among CAVI, Skin AF and Various Clinical Parameters |
Multivariate analysis and receiver-operating characteristic curve
Table 3 summarizes the results of a multiple regression analysis with CAVI or skin AF as a subordinate factor. Explanatory factors were selected by significant variables in univariate analysis. Age, skin AF, smoker, hs-CRP and HOMA-IR were identified as independent variables when CAVI was used as a subordinate factor. On the contrary, HOMA-IR, CAVI, hs-CRP and the smoker were identified as independent variables when skin AF was used as a subordinate factor. Figure 5 shows the receiver-operating characteristic curve analysis for the detection of high CAVI as ≥ 9.0 based on skin AF. The maximum Youden index indicated that skin AF of > 2.7 AU was the optimal cut-off point to determine the high CAVI (area under the curve = 0.718, P < 0.001) with true positive rate of 55.1% and false positive rate of 81.2%.
 Click to view | Table 3. Multiple Regression Analysis |
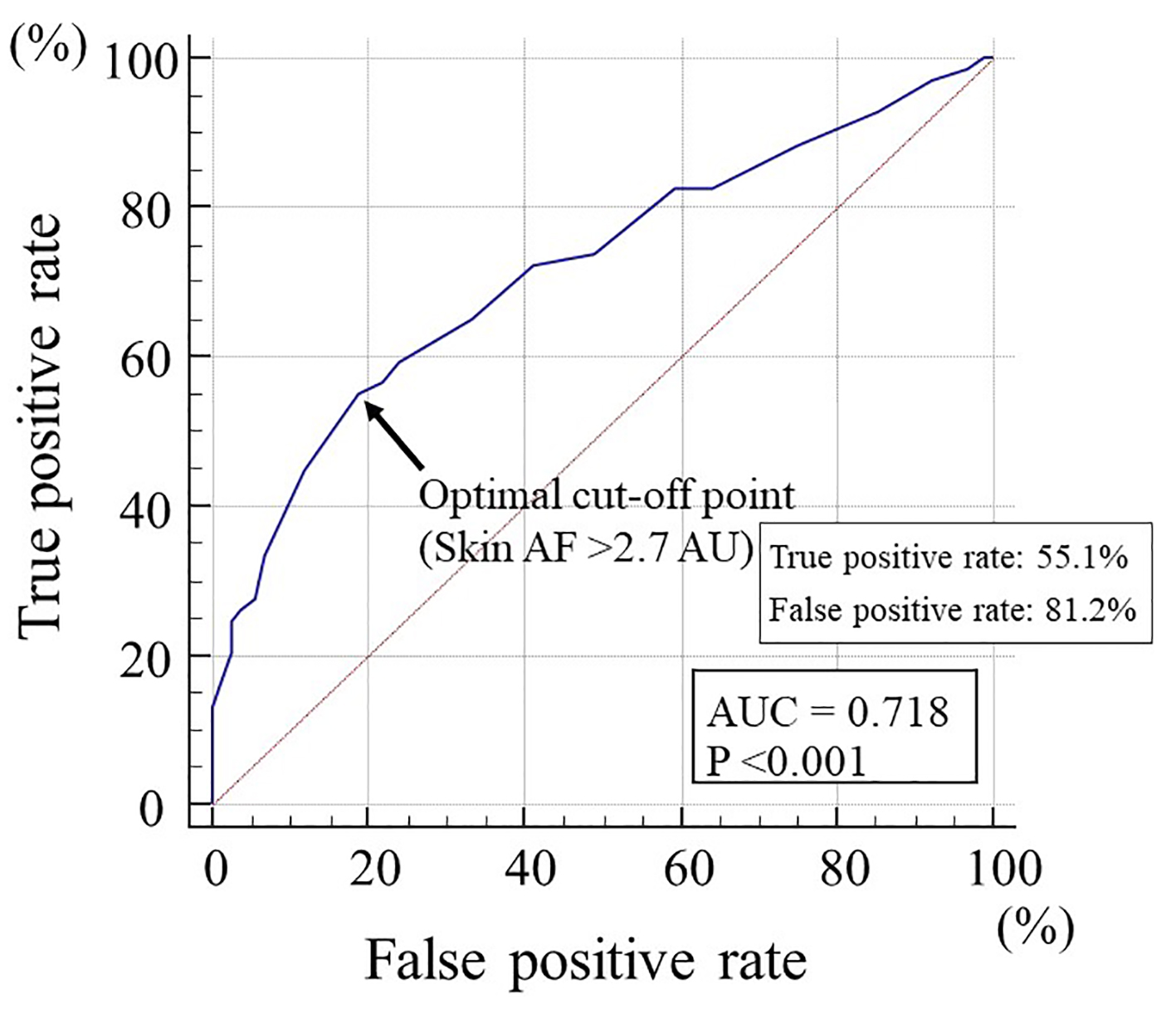 Click for large image | Figure 5. The receiver-operating characteristic curve analysis for the detection of high CAVI based on skin AF. The maximum Youden’s index indicated that skin AF of > 2.7 AU was the optimal cut-off point to determine the high CAVI of ≥ 9.0. AF: autofluorescence; CAVI: cardio-ankle vascular index; AU: arbitrary unit; AUC: area under the curve. |
| Discussion | ▴Top |
The results in this study indicated that skin AF was significantly positively correlated with CAVI; furthermore, multivariate analysis clarified that these two clinical parameters were independently associated. Previous studies reported that skin AF was significantly associated with physiological marker of arterial function including CAVI in patients with diabetes mellitus or kidney disease [10, 26-28]. In addition, this study also indicated that skin AF is considered as an important factor that affects the arterial function in patients with Mets. On the contrary, HOMA-IR as a marker of insulin resistance, smoking habits and hs-CRP as an inflammation marker were selected as independent variables for both CAVI and skin AF as a subordinate factor.
Previous studies have reported that AGEs or their receptors were associated with vascular cell calcification such as endothelial or smooth muscle cells or functional arterial aging [29-31]. In addition, the results in this study that indicate significant relationships between skin AF and CAVI can be interpreted as AGEs acting an important role for arterial function in patients with Mets. On the contrary, a number of studies indicated that insulin resistance acts a critical role in the pathogenesis of Mets. In addition, several researchers reported that insulin resistance affected arterial dysfunction [32, 33]. In this study, HOMA-IR was also significantly related with CAVI in patients with Mets, indicating that insulin resistance also acts an important role for arterial dysfunction. On the contrary, some basic studies indicated that AGEs affected insulin resistance through the several pathways such as AGEs receptor-1, sirtuin-1 and phosphoinositide 3-kinase [34, 35]. The results in this study also indicated that HOMA-IR was selected as one of the strongest variables for skin AF as a subordinate factor. Therefore, an intervention therapy was aggressively performed to reduce AGEs; consequently, CVD events were reduced in patients with Mets by improving insulin resistance.
Smoking habits have been known as one of the important risk factors for CVD incidence. In addition, several studies indicated that complication of smoking habits and Mets was significantly associated with higher CVD incidence risk [36]. The results in this study indicate a significant association between smoking habits and CAVI, which can also be interpreted that smoking habits were important target factors to promote arterial dysfunction in patients with Mets. On the contrary, several studies reported that the smoke can affect AGEs production [37, 38]. The results in this study also indicated that smoking habits or degree of smoking exposure were significantly associated with increased skin AF levels in patients with Mets. Thus, the results in previous and this study indicated that smoking cessation was strongly recommended for arterial function or AGEs production in patients with Mets.
Researchers emphasized that low-grade inflammation in the arterial wall plays a crucial role in the pathogenesis of atherosclerosis progression [39]. Indeed, several clinical studies have reported significant associations between inflammatory markers and CAVI [40, 41]. This study also indicated that hs-CRP was selected as independent variables for CAVI as a subordinate factor, indicating that inflammation acts an important role to promote arterial dysfunction in patients with Mets. On the contrary, several studies emphasized that AGEs or their receptors were associated with inflammation in Mets [42, 43]. This study also indicated that skin AF was significantly associated with hs-CRP in patients with Mets. On the contrary, medications such as statin or angiotensin receptor blocker were reported to decrease inflammation in the arterial wall [44, 45]. In addition, these medications also decreased AGE levels [46, 47]. In this study, all patients had no medications. Therefore, these medications, that is, statin or angiotensin receptor blocker, were aggressively administered on patients with Mets with high skin AF levels, which consequently possibly decrease CVD events by decreasing inflammation and AGE levels.
The target cut-off levels of skin AF should be determined in order to predict abnormal CAVI levels in clinical settings. This study clarifies the clinical usefulness of assessing skin AF to detect CAVI as high as ≥ 9.0, which is shown to be discriminatory of vascular failure by CAVI [48]. Receiver-operating characteristic curve analysis indicated that skin AF of > 2.7 AU is the optimal cut-off point to discriminate high CAVI. Several cut-off levels of skin AF for CVD risk were reported [14, 49, 50]. Although true positive rate was relatively low, this study indicated that CVD events may be decreased in patients with Mets to maintain skin AF of ≤ 2.7 AU. Lifestyle modification is undoubtedly one of the most important therapies to manage patients with Mets. In addition, Isami et al also reported that lifestyle habits such as physical activity, nonsmoking, adequate sleep, low mental stress level, eating breakfast and abstaining from sugary foods were independently associated with lower skin AF levels [51]. Therefore, good lifestyle habits are important to maintain lower skin AF levels in patients with Mets.
Limitations
This study had several limitations. First, a substantial number of patients did not undergo modalities, such as angiography, computed tomography, magnetic resonance imaging and echocardiography; therefore, asymptomatic CVD may have remained undetected. Second, female patients were not included in this study. A previous study indicated that the proportion of patients with Mets diagnosed according to the Japanese criteria in the general Japanese female population is extremely small compared to the male population (incidence: 12.1% in males, 1.7% in females) [52]. Therefore, Mets in females is very difficult to analyze in a single-unit range. The relationship between skin AF and CAVI in the Japanese female Mets population will be discussed in large-scale studies. Finally, this was single-center cross-sectional study, and the sample size was relatively small. An additional large number of studies, including evaluations of interventional therapies, are required to clarify the clinical significance of skin AF as a risk factor for CVD in patients with Mets.
Conclusions
In conclusion, this study indicates that skin AF was independently associated with CAVI, indicating that skin AF may be an important CVD risk factor in Japanese male patients with Mets. In addition, the risk value of skin AF was considered as higher than 2.7 AU. Further investigations in a large number of prospective studies, including intervention therapies, are required to validate the results in this study.
Acknowledgments
The author is grateful to the individuals who participated in this study.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
All patients provided informed consent.
Author Contributions
The author was involved in preparing the study design as well as acquisition, analysis and interpretation of data.
| References | ▴Top |
- Ford ES, Giles WH, Dietz WH. Prevalence of the metabolic syndrome among US adults: findings from the third National Health and Nutrition Examination Survey. JAMA. 2002;287(3):356-359.
doi pubmed - Ansarimoghaddam A, Adineh HA, Zareban I, Iranpour S, HosseinZadeh A, Kh F. Prevalence of metabolic syndrome in Middle-East countries: Meta-analysis of cross-sectional studies. Diabetes Metab Syndr. 2018;12(2):195-201.
doi pubmed - Tadaki S, Sakata Y, Miura Y, Miyata S, Asakura M, Shimada K, Yamamoto T, et al. Prognostic impacts of metabolic syndrome in patients with chronic heart failure- a multicenter prospective cohort study. Circ J. 2016;80(3):677-688.
doi pubmed - Galassi A, Reynolds K, He J. Metabolic syndrome and risk of cardiovascular disease: a meta-analysis. Am J Med. 2006;119(10):812-819.
doi pubmed - Mottillo S, Filion KB, Genest J, Joseph L, Pilote L, Poirier P, Rinfret S, et al. The metabolic syndrome and cardiovascular risk a systematic review and meta-analysis. J Am Coll Cardiol. 2010;56(14):1113-1132.
doi pubmed - Shirai K, Hiruta N, Song M, Kurosu T, Suzuki J, Tomaru T, Miyashita Y, et al. Cardio-ankle vascular index (CAVI) as a novel indicator of arterial stiffness: theory, evidence and perspectives. J Atheroscler Thromb. 2011;18(11):924-938.
doi pubmed - Endo K, Saiki A, Ohira M, Miyashita Y, Shirai K. Cardio-ankle vascular index may reflect endothelial function in type 2 diabetes. Int J Clin Pract. 2011;65(11):1200-1201.
doi pubmed - Liu J, Liu H, Zhao H, Shang G, Zhou Y, Li L, Wang H. Descriptive study of relationship between cardio-ankle vascular index and biomarkers in vascular-related diseases. Clin Exp Hypertens. 2017;39(5):468-472.
doi pubmed - Matsushita K, Ding N, Kim ED, Budoff M, Chirinos JA, Fernhall B, Hamburg NM, et al. Cardio-ankle vascular index and cardiovascular disease: Systematic review and meta-analysis of prospective and cross-sectional studies. J Clin Hypertens (Greenwich). 2019;21(1):16-24.
doi pubmed - Hitsumoto T. Clinical Significance of Cardio-Ankle Vascular index as a cardiovascular risk factor in elderly patients with type 2 diabetes mellitus. J Clin Med Res. 2018;10(4):330-336.
doi pubmed - Hitsumoto T, Shirai K. Factors affecting high-sensitivity cardiac troponin T elevation in Japanese metabolic syndrome patients. Diabetes Metab Syndr Obes. 2015;8:157-162.
doi pubmed - Laucevicius A, Ryliskyte L, Balsyte J, Badariene J, Puronaite R, Navickas R, Solovjova S. Association of cardio-ankle vascular index with cardiovascular risk factors and cardiovascular events in metabolic syndrome patients. Medicina (Kaunas). 2015;51(3):152-158.
doi pubmed - Furuya F, Shimura H, Takahashi K, Akiyama D, Motosugi A, Ikegishi Y, Haraguchi K, et al. Skin autofluorescence is a predictor of cardiovascular disease in chronic kidney disease patients. Ther Apher Dial. 2015;19(1):40-44.
doi pubmed - Noordzij MJ, Mulder DJ, Oomen PH, Brouwer T, Jager J, Castro Cabezas M, Lefrandt JD, et al. Skin autofluorescence and risk of micro- and macrovascular complications in patients with Type 2 diabetes mellitus-a multi-centre study. Diabet Med. 2012;29(12):1556-1561.
doi pubmed - Hitsumoto T. Clinical Significance of Skin Autofluorescence in Patients With Type 2 Diabetes Mellitus With Chronic Heart Failure. Cardiol Res. 2018;9(2):83-89.
doi pubmed - van Waateringe RP, Slagter SN, van Beek AP, van der Klauw MM, van Vliet-Ostaptchouk JV, Graaff R, Paterson AD, et al. Skin autofluorescence, a non-invasive biomarker for advanced glycation end products, is associated with the metabolic syndrome and its individual components. Diabetol Metab Syndr. 2017;9:42.
doi pubmed - Matsuzawa Y. Metabolic syndrome - definition and diagnostic criteria in Japan. J Atheroscler Thromb. 2005;12(6):301.
doi pubmed - Hitsumoto T, Takahashi M, Iizuka T, Shirai K. Relationship between metabolic syndrome and early stage coronary atherosclerosis. J Atheroscler Thromb. 2007;14(6):294-302.
doi pubmed - Meerwaldt R, Links TP, Graaff R, Hoogenberg K, Lefrandt JD, Baynes JW, Gans RO, et al. Increased accumulation of skin advanced glycation end-products precedes and correlates with clinical manifestation of diabetic neuropathy. Diabetologia. 2005;48(8):1637-1644.
doi pubmed - Nomoto K, Yagi M, Arita S, Hamada U, Yonei Y. A survey of fluorescence derived from advanced glycation end products in the skin of Japanese: differences with age and measurement location. Anti Aging Med. 2012;9(5):119-124.
- Meerwaldt R, Graaff R, Oomen PHN, Links TP, Jager JJ, Alderson NL, Thorpe SR, et al. Simple non-invasive assessment of advanced glycation endproduct accumulation. Diabetologia. 2004;47(7):1324-1330.
doi pubmed - Shirai K, Utino J, Otsuka K, Takata M. A novel blood pressure-independent arterial wall stiffness parameter; cardio-ankle vascular index (CAVI). J Atheroscler Thromb. 2006;13(2):101-107.
doi pubmed - Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem. 1972;18(6):499-502.
- Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28(7):412-419.
doi pubmed - Schisterman EF, Perkins NJ, Liu A, Bondell H. Optimal cut-point and its corresponding Youden Index to discriminate individuals using pooled blood samples. Epidemiology. 2005;16(1):73-81.
doi pubmed - Osawa S, Katakami N, Kuroda A, Takahara M, Sakamoto F, Kawamori D, Matsuoka T, et al. Skin autofluorescence is associated with early-stage atherosclerosis in patients with type 1 diabetes. J Atheroscler Thromb. 2017;24(3):312-326.
doi pubmed - Hitsumoto T. Clinical usefulness of the cardio-ankle vascular index as a predictor of primary cardiovascular events in patients with chronic kidney disease. J Clin Med Res. 2018;10(12):883-890.
doi pubmed - Wang CC, Wang YC, Wang GJ, Shen MY, Chang YL, Liou SY, Chen HC, et al. Skin autofluorescence is associated with endothelial dysfunction in uremic subjects on hemodialysis. PLoS One. 2016;11(1):e0147771.
doi pubmed - Yan SF, Ramasamy R, Schmidt AM. The RAGE axis: a fundamental mechanism signaling danger to the vulnerable vasculature. Circ Res. 2010;106(5):842-853.
doi pubmed - Grossin N, Auger F, Niquet-Leridon C, Durieux N, Montaigne D, Schmidt AM, Susen S, et al. Dietary CML-enriched protein induces functional arterial aging in a RAGE-dependent manner in mice. Mol Nutr Food Res. 2015;59(5):927-938.
doi pubmed - Suga T, Iso T, Shimizu T, Tanaka T, Yamagishi S, Takeuchi M, Imaizumi T, et al. Activation of receptor for advanced glycation end products induces osteogenic differentiation of vascular smooth muscle cells. J Atheroscler Thromb. 2011;18(8):670-683.
doi pubmed - Webb DR, Khunti K, Silverman R, Gray LJ, Srinivasan B, Lacy PS, Williams B, et al. Impact of metabolic indices on central artery stiffness: independent association of insulin resistance and glucose with aortic pulse wave velocity. Diabetologia. 2010;53(6):1190-1198.
doi pubmed - Fakhrzadeh H, Sharifi F, Alizadeh M, Arzaghi SM, Tajallizade-Khoob Y, Tootee A, Alatab S, et al. Relationship between insulin resistance and subclinical atherosclerosis in individuals with and without type 2 diabetes mellitus. J Diabetes Metab Disord. 2015;15:41.
doi pubmed - Uribarri J, Cai W, Ramdas M, Goodman S, Pyzik R, Chen X, Zhu L, et al. Restriction of advanced glycation end products improves insulin resistance in human type 2 diabetes: potential role of AGER1 and SIRT1. Diabetes Care. 2011;34(7):1610-1616.
doi pubmed - Miele C, Riboulet A, Maitan MA, Oriente F, Romano C, Formisano P, Giudicelli J, et al. Human glycated albumin affects glucose metabolism in L6 skeletal muscle cells by impairing insulin-induced insulin receptor substrate (IRS) signaling through a protein kinase C alpha-mediated mechanism. J Biol Chem. 2003;278(48):47376-47387.
doi pubmed - Higashiyama A, Okamura T, Ono Y, Watanabe M, Kokubo Y, Okayama A. Risk of smoking and metabolic syndrome for incidence of cardiovascular disease—comparison of relative contribution in urban Japanese population: the Suita study. Circ J. 2009;73(12):2258-2263.
doi pubmed - Cerami C, Founds H, Nicholl I, Mitsuhashi T, Giordano D, Vanpatten S, Lee A, et al. Tobacco smoke is a source of toxic reactive glycation products. Proc Natl Acad Sci U S A. 1997;94(25):13915-13920.
doi pubmed - Nicholl ID, Bucala R. Advanced glycation endproducts and cigarette smoking. Cell Mol Biol (Noisy-le-grand). 1998;44(7):1025-1033.
- Geovanini GR, Libby P. Atherosclerosis and inflammation: overview and updates. Clin Sci (Lond). 2018;132(12):1243-1252.
doi pubmed - Kotani K, Yamada T, Miyamoto M, Kario K, Ishibashi S, Taniguchi N. The correlation between the cardio-ankle vascular index (CAVI) and serum amyloid A in asymptomatic Japanese subjects. Heart Vessels. 2012;27(5):499-504.
doi pubmed - Higashiyama A, Wakabayashi I, Kubota Y, Adachi Y, Hayashibe A, Nishimura K, Sugiyama D, et al. Does high-sensitivity C-reactive protein or low-density lipoprotein cholesterol show a stronger relationship with the cardio-ankle vascular index in healthy community dwellers?: the KOBE study. J Atheroscler Thromb. 2012;19(11):1027-1034.
doi pubmed - Uribarri J, Cai W, Woodward M, Tripp E, Goldberg L, Pyzik R, Yee K, et al. Elevated serum advanced glycation endproducts in obese indicate risk for the metabolic syndrome: a link between healthy and unhealthy obesity? J Clin Endocrinol Metab. 2015;100(5):1957-1966.
doi pubmed - Momma H, Niu K, Kobayashi Y, Huang C, Chujo M, Otomo A, Tadaura H, et al. Higher serum soluble receptor for advanced glycation end product levels and lower prevalence of metabolic syndrome among Japanese adult men: a cross-sectional study. Diabetol Metab Syndr. 2014;6(1):33.
doi pubmed - Pirro M, Simental-Mendia LE, Bianconi V, Watts GF, Banach M, Sahebkar A. Effect of statin therapy on arterial wall inflammation based on 18F-FDG PET/CT: a systematic review and meta-analysis of interventional studies. J Clin Med. 2019;8(1):118.
doi pubmed - Fujiwara Y, Shiraya S, Miyake T, Yamakawa S, Aoki M, Makino H, Nishimura M, et al. Inhibition of experimental abdominal aortic aneurysm in a rat model by the angiotensin receptor blocker valsartan. Int J Mol Med. 2008;22(6):703-708.
- Mukai H, Svedberg O, Lindholm B, Dai L, Heimburger O, Barany P, Anderstam B, et al. Skin autofluorescence, arterial stiffness and Framingham risk score as predictors of clinical outcome in chronic kidney disease patients: a cohort study. Nephrol Dial Transplant. 2019;34(3):442-448.
doi pubmed - Forbes JM, Thomas MC, Thorpe SR, Alderson NL, Cooper ME. The effects of valsartan on the accumulation of circulating and renal advanced glycation end products in experimental diabetes. Kidney Int. 2004;66(Suppl 92):S105-S107.
doi pubmed - Tanaka A, Tomiyama H, Maruhashi T, Matsuzawa Y, Miyoshi T, Kabutoya T, Kario K, et al. Physiological diagnostic criteria for vascular failure. Hypertension. 2018;72(5):1060-1071.
doi pubmed - Lutgers HL, Graaff R, Links TP, Ubink-Veltmaat LJ, Bilo HJ, Gans RO, Smit AJ. Skin autofluorescence as a noninvasive marker of vascular damage in patients with type 2 diabetes. Diabetes Care. 2006;29(12):2654-2659.
doi pubmed - Wang CC, Wang YC, Wang GJ, Shen MY, Chang YL, Liou SY, Chen HC, et al. Skin autofluorescence is associated with inappropriate left ventricular mass and diastolic dysfunction in subjects at risk for cardiovascular disease. Cardiovasc Diabetol. 2017;16(1):15.
doi pubmed - Isami F, West BJ, Nakajima S, Yamagishi SI. Association of advanced glycation end products, evaluated by skin autofluorescence, with lifestyle habits in a general Japanese population. J Int Med Res. 2018;46(3):1043-1051.
doi pubmed - Arai H, Yamamoto A, Matsuzawa Y, Saito Y, Yamada N, Oikawa S, Mabuchi H, et al. Prevalence of metabolic syndrome in the general Japanese population in 2000. J Atheroscler Thromb. 2006;13(4):202-208.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


