| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Original Article
Volume 15, Number 1, February 2024, pages 56-66
Improving Respiratory Muscle Strength and Overall Function in Patients With Cardiovascular Disease Through Rehabilitation Hospitals
Tomohiro Matsuoa, Tomoyuki Morisawab, d, Takuro Ohtsuboa, Katsuhiro Uenoa, Shuichi Kozawac
aDepartment of Rehabilitation, Nishi Memorial Port-island Rehabilitation Hospital, Chuo-ku, Kobe, Hyogo 650-0046, Japan
bDepartment of Physical Therapy, Faculty of Health Sciences, Juntendo University, Bunkyo-ku, Tokyo 113-8421, Japan
cDepartment of Cardiology, Nishi Memorial Port-island Rehabilitation Hospital, Chuo-ku, Kobe, Hyogo 650-0046, Japan
dCorresponding Author: Tomoyuki Morisawa, Department of Physical Therapy, Faculty of Health Sciences, Juntendo University, Bunkyo-ku, Tokyo 113-8421, Japan
Manuscript submitted January 13, 2024, accepted February 21, 2024, published online February 28, 2024
Short title: Improving RMS on CR in Rehab Hospitals
doi: https://doi.org/10.14740/cr1616
| Abstract | ▴Top |
Background: The prevalence of respiratory sarcopenia and its effect on respiratory muscle strength (RMS) in patients with cardiovascular disease (CVD), who are transferred to a convalescent rehabilitation hospital after acute care and require continuous cardiac rehabilitation (CR), is currently unclear. This study aimed to assess changes in RMS, physical function, and activities of daily living (ADL) before and after CR performed in a rehabilitation hospital.
Methods: Of 50 consecutive patients transferred to a rehabilitation hospital for ongoing CR, 30 fulfilled the inclusion criteria. Maximal inspiratory and expiratory pressures (MIP and MEP, respectively) were measured at transfer, and patients with decreased RMS were diagnosed with respiratory sarcopenia. RMS, physical function, exercise tolerance, ADL ability, and health-related quality of life (HR-QoL) were measured and compared at transfer and discharge.
Results: The prevalence of respiratory sarcopenia at the time of transfer to the rehabilitation hospital was 93.3%. RMS assessments at transfer and discharge demonstrated significant improvements in %MIP (from 46.3±26.1% to 63.6±33.7%) and %MEP (from 44.8±17.3% to 56.6±21.8%). Short physical performance battery, gait speed, handgrip strength, and knee extension muscle strength significantly improved, along with significant prolongation of 6-min walking distance as a measure of exercise tolerance. ADL assessment using the functional independence measure revealed significant improvement, as did HR-QoL assessed according to the five-dimension, five-level, EuroQoL instrument, following CR.
Conclusions: Although respiratory sarcopenia was highly prevalent among patients with CVD who required transfer to a rehabilitation hospital after acute care, continuous CR significantly improved RMS, ADL, physical function, and exercise tolerance. These findings support the continued expansion of CR, particularly in dedicated rehabilitation hospitals.
Keywords: Rehabilitation hospital; Cardiac rehabilitation; Respiratory muscle strength
| Introduction | ▴Top |
Advances in acute-care cardiovascular medicine have made it feasible to treat elderly and critically ill patients who were previously difficult to manage. However, these patients often encounter delays in achieving acute rehabilitation goals owing to the complexity of their disabilities and the extended recovery time resulting from preexisting conditions such as sarcopenia, frailty, and multimorbidity. Hospital-acquired functional decline (HAD) affects approximately 20% of elderly patients after acute care [1], and 10-20% of elderly patients with cardiovascular disease (CVD) experience HAD [2-4]. For these individuals, convalescent rehabilitation hospitals play a crucial role in providing continued intensive inpatient cardiac rehabilitation (CR) as an integral component of phase 2 CR to enhance their physical function and activities of daily living (ADL). Although CR in rehabilitation hospitals has been shown to significantly improve physical function, exercise tolerance, ADL ability, and health-related quality of life (HR-QoL) in patients with CVD [5, 6], its effect on respiratory muscle strength (RMS), a prognostic factor for patients with CVD [7, 8], remains unclear. In particular, the recent inclusion of CVDs in the calculation requirements for convalescent rehabilitation in Japan’s FY 2022 Medical Fee Revision underscores the urgency to understand the current state of care. This amendment highlights the growing recognition of the importance of specialized rehabilitation services for patients with CVD and the need for empirical data to guide these services’ optimization.
In recent years, the age-related loss of RMS and muscle mass has been referred to as “respiratory sarcopenia” [9, 10], defined as whole-body sarcopenia and low respiratory muscle mass followed by a decrease in RMS and/or respiratory function [10], and has attracted attention as a new therapeutic target in the elderly. Morisawa et al [11] reported that respiratory sarcopenia may be present in approximately one-half of community-dwelling elderly individuals; however, respiratory sarcopenia is more likely to be a comorbidity among elderly patients with CVD. Patients with CVD are prone to dyspnea during exercise, leading to exercise intolerance and reduced quality of life [12, 13]. This exercise-induced dyspnea is partly due to reduced RMS in approximately 30-50% of patients with CVD [13, 14]. Additionally, patients with CVD exhibit reduced RMS independent of their underlying cardiac condition, such as heart failure (HF), myocardial infarction, or postoperative cardiovascular surgery [15-17], and evaluation of RMS function as part of phase 2 CR has attracted attention [18]. Such assessments are crucial for understanding the extent of exercise intolerance and diminished quality of life among patients with CVD, which is frequently exacerbated by reduced RMS.
This study is therefore focused on elucidating the prevalence of respiratory sarcopenia among patients with CVD requiring continuous CR in a convalescent rehabilitation hospital, and on evaluating the impact of CR interventions on RMS alterations. Given the critical role of RMS in the overall health and rehabilitation outcomes of patients with CVD, our research seeks to bridge the gap in knowledge regarding the effectiveness of CR in addressing respiratory sarcopenia, underlining the necessity for routine RMS evaluations as a component of comprehensive CR efforts.
| Materials and Methods | ▴Top |
Study design and study population
This retrospective cohort study was conducted at a single center between February 2022 and June 2023. The study included consecutive patients treated for CVD at an acute care hospital, who were subsequently transferred to the Nishi Memorial Port-island Rehabilitation Hospital (Hyogo, Japan) due to inadequate recovery of physical function and ADL, necessitating continued CR in a convalescent rehabilitation hospital. Exclusion criteria included the following: age < 18 years; mobility impairments (e.g., cerebrovascular disease, quadriplegia, hemiplegia, or spinal cord infarction); cognitive impairments that hindered accurate measurements; unsuitability for RMS testing (e.g., pneumothorax or conservative treatment after acute aortic dissection); and refusal to participate in the study. Data from patients who died during hospitalization or developed a newly occurring cerebrovascular disease were also excluded. This study was approved by the Ethics Committee of Nishi Memorial Port-island Rehabilitation Hospital (approval No. 18), and written informed consent was obtained from each patient. The study adhered to the ethical standards for human participant research in accordance with the Declaration of Helsinki.
Data collection
Baseline characteristics extracted from medical records and interviews included age, sex, body mass index, pre-admission ADL (Barthel Index), pre-admission life function (Kihon Checklist (KCL)), presence of a caregiver, primary diseases in the acute care hospital (HF, postoperative cardiovascular surgery, postoperative aortic surgery, postoperative combined cardiovascular and aortic surgery), and length of stay in the acute care hospital. The KCL, a 25-question yes/no questionnaire, was used to assess the severity of frailty [19]. It is stratified into three levels: robust (0 to 3 points); pre-frailty (4 to 7 points); and frailty (≥ 8 points) [20]. Additional data collected included New York Heart Association functional classification, comorbidities, echocardiographic findings, and biochemical test results at the time of transfer to a rehabilitation hospital. The total CR time per day, length of hospital stays, and patient outcomes in the rehabilitation hospital were also recorded.
Determination of respiratory sarcopenia
Respiratory sarcopenia is typically identified based on a combination of decreased respiratory muscle mass and RMS according to existing diagnostic criteria [10, 21]. Standard practices involve measuring diaphragm muscle mass using computed tomography (CT) or ultrasound. Due to the practical limitations in the setting of Japanese convalescent rehabilitation hospitals, where routine CT or ultrasound evaluations for RMS are not commonly performed, we were unable to directly assess RMS through these imaging techniques in any of the patients included in our study. When these methods are impractical, skeletal muscle mass measurement using bioelectrical impedance analysis (BIA) is an alternative [10]. In this study, many participants had pacemakers, which precluded the use of BIA for muscle mass measurements. Therefore, participants exhibiting decreased RMS were categorized as “possible respiratory sarcopenia” [21]. Respiratory muscle weakness was defined as an RMS value < 70% of the reference standard. This threshold was based on previous research [22, 23] and was used to identify potential cases of respiratory sarcopenia when direct measurement of muscle mass was not feasible.
Assessments of RMS
RMS was measured using an RMS tester (IOP-01, Kobata Keiki Co., Osaka, Japan). Maximal inspiratory and expiratory efforts were performed twice with subjects in the sitting position, with the highest values recorded for maximal inspiratory pressure (MIP) and maximal expiratory pressure (MEP). Predicted MIP and MEP values were calculated using age, sex, and body size-adjusted equations (predicted MIP (male) = 45.0 - 0.74 × age + 0.27 × height + 0.60 × weight; predicted MIP (female) = -1.5 - 0.41 × age + 0.48 × height + 0.12 × weight. Predicted MEP (male) = 25.1 - 0.37 × age + 0.20 × height + 1.20 × weight; predicted MEP (female) = -19.1 - 0.18 × age + 0.43 × height + 0.56 × weight) [24]. %MIP and %MEP were calculated by dividing the actual MIP and MEP by the predicted MIP and MEP, respectively, and multiplied by 100.
ADL, physical function, and exercise tolerance assessments
ADLs were evaluated by trained physical and occupational therapists using the functional independence measure (FIM) [25] on transfer to a rehabilitation hospital and discharge. The FIM is used to assess the degree to which an individual is independent or requires assistance to perform various motor and cognitive ADL. The FIM consists of 18 items, including 13 motor and five cognitive items, and is scored on a 7-point Likert scale ranging from “completely dependent on assistance (score, 1)” to “completely independent (score, 7)”. Lower scores indicate lower independence in ADL.
Physical function was measured at transfer and discharge using the short physical performance battery (SPPB), handgrip strength, knee extension muscle strength, and gait speed. The SPPB is a reliable and valid battery for assessing physical function in older individuals [26], calculated from balance ability, 4 m walk time, and 5-repetition sit-to-stand time according to the method described by Guralnik et al [27]. Handgrip strength was measured twice bilaterally using a digital handgrip gauge (T-2177; TOEI LIGHT Co., Ltd., Shizuoka, Japan), and the maximum value was recorded. Knee extension muscle strength was assessed using a handheld dynamometer (µTas F-1, Anima Co., Ltd., Japan). Briefly, isometric knee extension strength was measured with the knee joint flexed 90° in the sitting position and then extended with maximal effort. Knee extension muscle strength was measured twice, the results were divided by body weight (BW) to calculate %BW, and the maximum value was used. Gait speed was measured at a comfortable walking speed with an aided walking path (10 m in length) and converted to m/s. Exercise tolerance was assessed by the 6-min walking distance (6MD) using the 6-min walk test (6MWT) at transfer and discharge. The 6MWT consisted of walking a straight line for 20 m in a rehabilitation room with no steps or obstacles, and the maximum distance each subject could walk in 6 min, according to the standards of the American Thoracic Society [28], with appropriate instructions given before the test.
Cognitive function and HR-QoL assessments
Cognitive function was assessed in accordance with the Japanese version of the mini-mental state examination (MMSE) [29], with scores ranging from 0 to 30, and higher scores indicating better cognitive function. HR-QoL was measured in accordance with the Japanese version of the five-dimension, five-level, EuroQoL instrument (EQ-5D-5L) questionnaire [30], with scores calculated using the EQ-5D-5L index value calculator version 2.0 [31].
CR
All patients transferred to the rehabilitation hospital underwent physical and occupational therapies as prescribed by their physicians. Exercise programs in CR, adhering to Japanese Circulation Society guidelines [32], focus on aerobic exercise, resistance training, and stretching [33]. Although cardiopulmonary exercise testing based on anaerobic thresholds is recommended to determine exercise intensity [33], this is often not feasible for patients transferred to a rehabilitation hospital due to significant decline in ADL, coexisting stroke or musculoskeletal disease. Therefore, we primarily used the Borg Scale [34] to assess perceived exertion and the Talk Test to monitor relative intensity [35]. The intensity and duration of exercise were increased or decreased in response to changes in the patient’s medical condition. In addition, when necessary, rehabilitation included breathing exercises, thoracic stretching, and accessory respiratory muscle conditioning tailored to patient condition. Breathing exercises were performed when deemed necessary by the treating therapist, with an emphasis on deep breathing exercises, with the physiotherapist instructing the rhythm of inhalation and exhalation and adjusting the speed and depth of inhalation and exhalation. Individualized rehabilitation programs incorporated ADL and balance training, exercise instruction, and patient education and were provided daily for up to 3 h per day. Educational sessions included exercise, nutrition, and stress management and were tailored to the patient’s post-discharge lifestyle. Speech therapy was prescribed when deemed necessary in the presence of swallowing difficulties.
Statistical analysis
The prevalence of respiratory sarcopenia upon transfer to the rehabilitation hospital was calculated as the percentage of patients presenting with respiratory sarcopenia at that time. Paired t-test and the Wilcoxon signed-rank test were used to compare various parameters, including RMS, before and after intervention at the rehabilitation hospital. Statistical analyses were performed using SPSS version 29.0 (IBM Corp., Armonk, NY, USA); differences with P < 0.05 were considered to be statistically significant.
| Results | ▴Top |
Patient demographics
Of the 50 consecutive patients transferred to the convalescent rehabilitation hospital following acute CVD treatment, 30 fulfilled the inclusion criteria after applying the exclusion criteria such as mobility issues and RMS measurement challenges. A flow-diagram illustrating study participant inclusion/exclusion is presented in Figure 1, and basic characteristics are summarized in Table 1.
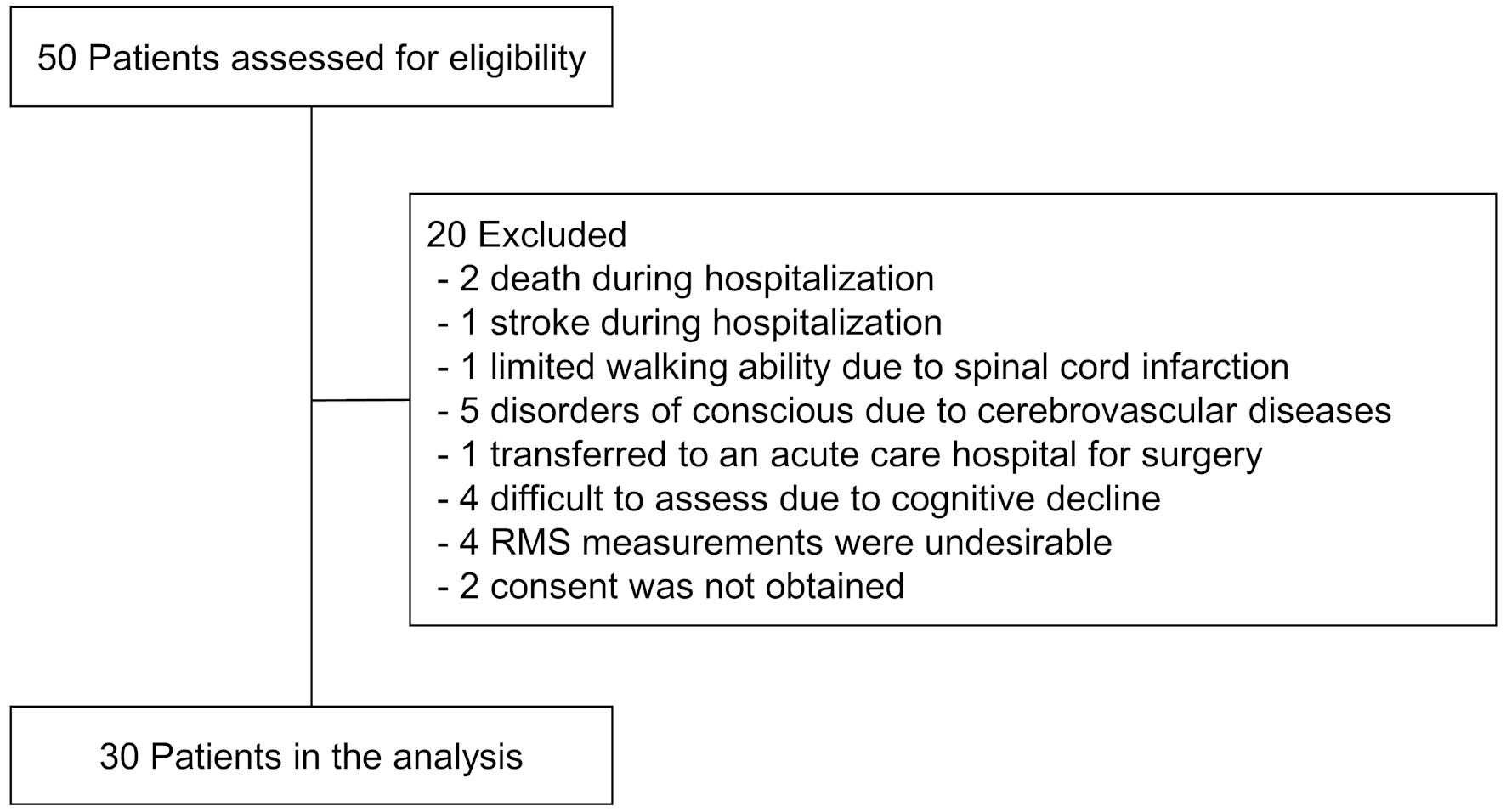 Click for large image | Figure 1. Flowchart of participants throughout the study. RMS: respiratory muscle strength. |
 Click to view | Table 1. Baseline Characteristics of the Patients |
Prevalence of respiratory sarcopenia and changes in RMS
On transfer to the rehabilitation hospital, the prevalence of respiratory sarcopenia was 93.3%, with the majority of patients exhibiting respiratory sarcopenia (Table 1). Assessment of RMS at transfer and discharge revealed significant improvements in mean (± standard deviation (SD)) %MIP, increasing from 46.3±26.1% to 63.6±33.7% (P < 0.001). Similarly, mean %MEP exhibited a significant improvement, from 44.8±17.3% to 56.6±21.8% (P < 0.001), in expiratory muscle strength (Fig. 2). The effect of recovery was not influenced by the presence or absence of beta-blocker therapy (Table 2).
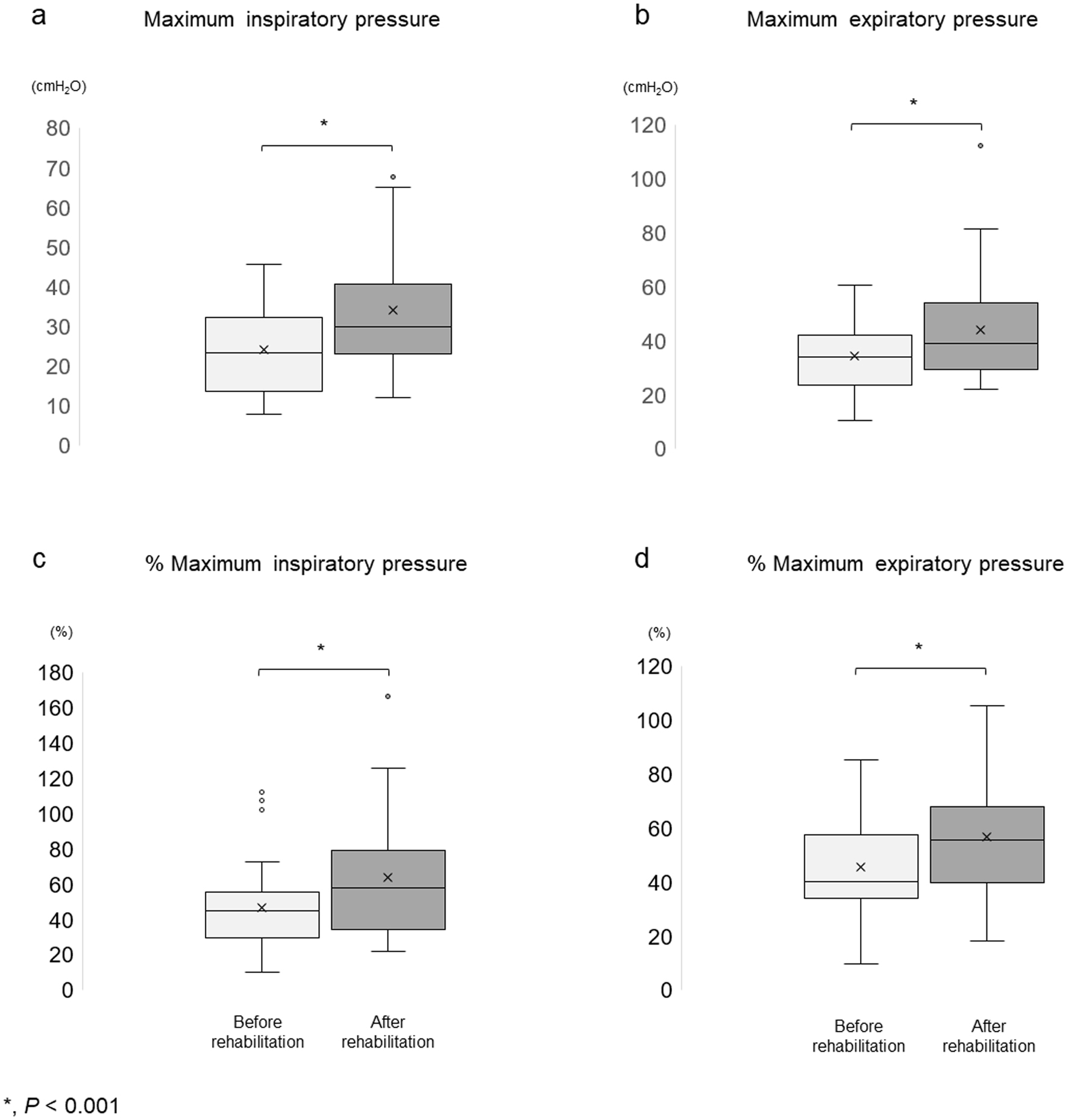 Click for large image | Figure 2. Changes in respiratory muscle strength. (a, b) Actual maximal inspiratory and expiratory pressure. (c, d) Calculated % maximal inspiratory and expiratory pressure. In the box-and-whisker plot, the box represented the interquartile range (IQR), while the whiskers extended to the minimum and maximum values within 1.5 times the IQR. Data points outside this range were considered outliers and were represented by circles. |
 Click to view | Table 2. Change in Rehabilitation Outcome in the Patient on and off Beta-Blockers |
Change in ADL, physical function, exercise tolerance, cognitive function, and HR-QoL
ADL assessments at transfer and discharge exhibited significant improvements in total FIM score, motor items of the FIM, and cognitive items of the FIM (P < 0.001). Physical function significantly improved after CR, as evidenced by SPPB scores (P < 0.001), gait speed (P < 0.001), handgrip strength (P = 0.01), and knee extension muscle strength (P < 0.001). Exercise tolerance, as assessed according to 6MD, also significantly improved after CR at the rehabilitation hospital (P < 0.001). Furthermore, cognitive function, as measured using the MMSE, improved significantly (P = 0.038). HR-QoL significantly improved, as indicated by EQ-5D-5L scores (P < 0.001) (Table 3).
 Click to view | Table 3. Changes in ADL, Physical Function, Exercise Tolerance, Cognitive Function, and QoL After Rehabilitation |
Rehabilitation dose
The mean total rehabilitation time per day in the rehabilitation hospital was 132.5 ± 13.4 min, including patient education. The mean length of hospital stay was 65.9 ± 30.4 days, and most patients were discharged to home (Table 1).
| Discussion | ▴Top |
Results of the present study demonstrated the effectiveness of transferring patients to a rehabilitation hospital for continued CR in improving RMS, ADL, physical function, and exercise tolerance after treatment for acute CVD. To the best of our knowledge, this was the first investigation in Japan to address the effect of CR on RMS in patients with CVD in rehabilitation hospitals.
Our findings showed that most participants, primarily elderly females with a history of musculoskeletal disease or HF, benefited from continued CR. Such patients often present with frailty yet are independent in ADL before admission, it is likely that these patients require continued CR in a rehabilitation hospital rather than being discharged after a short stay in an acute care hospital. The importance of continued CR in a rehabilitation hospital is underscored by the observed enhancements in RMS and physical outcomes, aligning with previous research indicating diminished functional recovery and CR efficacy in similar patient populations.
With a focus on RMS, the mean RMS at transfer was less than one-half of the predicted values for both MIP and MEP. Upon transfer, only 10% of the patients exhibited inspiratory muscle strength equivalent to that of normal subjects, whereas all patients exhibited expiratory muscle strength lower than normal. It is well documented that RMS is reduced in patients with CVD, such as those with HF, following cardiac surgery [15, 16]. However, in patients with CVD requiring transfer to a rehabilitation hospital, inspiratory muscle strength was even lower than that reported in a previous study (60-68% of the predicted value) [16]. Nevertheless, RMS significantly improved both inspiration and expiration following continued CR at a convalescent rehabilitation hospital. Previous studies have reported significant improvements in inspiratory muscle strength in patients with HF after 12 weeks of aerobic exercise [36], and in both expiratory and inspiratory muscle strength after a 12-week, phase 2 CR program focusing on aerobic exercise and resistance training following coronary bypass surgery [37].
Additionally, an 8-week home-based walking program led to a 41% improvement in inspiratory muscle strength in patients with myocardial infarction [17]. Findings of the present study are consistent with these reports, suggesting that long-term outpatient CR can enhance RMS, possibly reflecting the effects of CR in rehabilitation hospitals. Furthermore, patients with inspiratory muscle weakness at baseline were more likely to exhibit an improvement in MIP after CR [38], and intensive, prolonged, inpatient CR in a rehabilitation hospital may be more effective in improving RMS. In addition, increased MIP with CR has been reported to be associated with lower rates of all-cause mortality and cardiovascular events [38]. Patients with CVD who have a decreased RMS after acute care may also have an improved prognosis if they are offered continued CR in a convalescent rehabilitation hospital. However, when RMS decline was defined as < 70% of the predicted MIP [22, 23], two-thirds of patients continued to experience respiratory muscle weakness at discharge from the rehabilitation hospital. Respiratory muscle weakness has been reported to be associated with increased all-cause mortality and rehospitalization for CVD [7, 8], and reconsideration of the CR program is warranted for these patients. There are anecdotal reports that RMS training, particularly inspiratory muscle training (IMT), in combination with conventional CR in patients with CVD, further improves RMS [37, 39, 40]. Therefore, the addition of IMT to the CR program may be considered in patients presenting at a rehabilitation hospital with significant respiratory muscle weakness. However, no additional benefit of IMT over conventional aerobic exercise alone has been reported in outpatient CR combining aerobic exercise with high-intensity IMT in patients after coronary artery bypass graft surgery [41]. Thus, it is uncertain whether introducing IMT in rehabilitation hospitals, where a more individualized approach to care is required, will be beneficial.
Physical function, exercise tolerance, cognitive function, ADL, and HR-QoL significantly improved with continued CR in the rehabilitation hospital. Regarding the effect of CR on physical function and exercise tolerance, the reported minimal clinically important difference [42-44] was significantly higher in the present investigation as well as in previous studies in rehabilitation hospitals [5, 6]. In addition, cognitive function significantly improved in the CR group at the rehabilitation hospital, despite the exclusion of the cognitively impaired group, for whom accurate testing was difficult. This may be due to the reported improvement in memory scores with exercise therapy in older patients [45] and rehabilitation, which includes cognitive communication through occupational and speech therapy. Considering that the median CR time during hospitalization in Japan in the report by Kanazawa et al [46] was approximately 30 min per day and the length of hospitalization was approximately 3 weeks, the average daily CR time in this study was approximately 130 min, and the fact that CR was performed intensively and for a prolonged period may have influenced improvement compared with previous studies. Furthermore, in the Rehabilitation Therapy in Older Acute Heart Failure Patients (REHAB-HF) trial [48], physical function and HR-QoL were significantly improved in hospitalized patients with acute HF after a transitional, tailored, and incremental rehabilitation intervention compared with those in the control group. This suggests that individualized CR intervention in a rehabilitation hospital may improve physical function and HR-QoL more than conventional CR in an acute care hospital.
These results suggest that transfer to a rehabilitation hospital and continued CR are useful not only for improving ADL and physical function but also for improving RMS. Future research should aim to determine the effect of RMS improvement on the prognosis of patients who continue CR in rehabilitation hospitals.
Study limitations
This study had several limitations. First, the number of patients in the long-term rehabilitation hospital was limited; as such, the sample size was correspondingly small. Second, Japanese rehabilitation hospitals have not been able to adequately diagnose respiratory sarcopenia according to previous studies because imaging and respiratory function tests are not commonly performed, and RMS could not be directly evaluated by these diagnostic techniques. Third, the small sample size did not enable us to analyze factors influencing improved RMS. Forth, it is difficult to assess pre-admission respiratory and physical functions. This difficulty is compounded by the inherent complexity in determining how much the assessments at the point of transfer to the rehabilitation facility are influenced by the patients’ conditions prior to admission. Finally, the lack of a control group significantly impacts the strength and generalizability of the findings. The absence of a control group precludes definitive conclusions regarding the causality of observed improvements in RMS, potentially attributing outcomes to the rehabilitation interventions without rigorous comparative analysis.
Conclusions
Respiratory sarcopenia was present in most patients with CVD who required a transfer to a rehabilitation hospital after undergoing acute care. However, continuous CR has been shown to significantly improve RMS, ADL, physical function, exercise tolerance, and HR-QoL. These findings support the further expansion of CR, which should be intensively practiced in rehabilitation hospitals.
Acknowledgments
The authors thank all staff members of the Nishi Memorial Port-island Rehabilitation Hospital.
Financial Disclosure
This study was funded by the Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (KAKENHI) (Grant No. 20K11193 to Tomoyuki Morisawa).
Conflict of Interest
The authors declare no conflict of interest with this manuscript.
Informed Consent
All patients provided written informed consent.
Author Contributions
TM (Tomohiro Matsuo): acquisition, analysis, and interpretation of data; writing and revising the manuscript; and composition of tables and figures. TM (Tomoyuki Morisawa): conception and design of the study, analysis, interpretation of data, and critical revision of the manuscript for important intellectual content. TO and KU contributed to data acquisition. SK critically revised the manuscript. All authors have approved the final version of the manuscript for publication.
Data Availability
Data supporting the findings of this study are available from the corresponding author upon reasonable request.
Abbreviations
HAD: hospitalization-associated disability; CVD: cardiovascular disease; CR: cardiac rehabilitation; ADL: activities of daily living; HR-QoL: health-related quality of life; RMS: respiratory muscle strength; HF: heart failure; KCL: Kihon Checklist; CT: computed tomography; MIP: maximal inspiratory pressure; MEP: maximal expiratory pressure; FIM: functional independence measure; SPPB: short physical performance battery; BW: body weight; 6MD: 6-min walking distance; 6MWT: 6-min walk test; MMSE: mini-mental state examination; EQ-5D-5L: EuroQoL five-dimension, five-level
| References | ▴Top |
- Loyd C, Markland AD, Zhang Y, Fowler M, Harper S, Wright NC, Carter CS, et al. Prevalence of hospital-associated disability in older adults: a meta-analysis. J Am Med Dir Assoc. 2020;21(4):455-461.e455.
doi pubmed pmc - Yaku H, Kato T, Morimoto T, Inuzuka Y, Tamaki Y, Ozasa N, Yamamoto E, et al. Risk factors and clinical outcomes of functional decline during hospitalisation in very old patients with acute decompensated heart failure: an observational study. BMJ Open. 2020;10(2):e032674.
doi pubmed pmc - Takabayashi K, Kitaguchi S, Iwatsu K, Morikami Y, Ichinohe T, Yamamoto T, Takenaka K, et al. A decline in activities of daily living due to acute heart failure is an independent risk factor of hospitalization for heart failure and mortality. J Cardiol. 2019;73(6):522-529.
doi pubmed - Morisawa T, Saitoh M, Otsuka S, Takamura G, Tahara M, Ochi Y, Takahashi Y, et al. Association between hospital-acquired functional decline and 2-year readmission or mortality after cardiac surgery in older patients: a multicenter, prospective cohort study. Aging Clin Exp Res. 2023;35(3):649-657.
doi pubmed - Morisawa T, Ueno K, Fukuda Y, Kanazawa N, Kawaguchi H, Zaiki R, Fuzisaki H, et al. Significance of sequential cardiac rehabilitation program through inter-hospital cooperation between acute care and rehabilitation hospitals in elderly patients after cardiac surgery in Japan. Heart Vessels. 2017;32(10):1220-1226.
doi pubmed - Matsuo T, Morisawa T, Yamamoto S, Nyuba K, Maehata M, Fujii D, Iwaki H, et al. Effect of in-patient cardiac rehabilitation at rehabilitation hospital for cardiology patients: two-center trial. Heart Vessels. 2024;39(1):75-85.
doi pubmed - Meyer FJ, Borst MM, Zugck C, Kirschke A, Schellberg D, Kubler W, Haass M. Respiratory muscle dysfunction in congestive heart failure: clinical correlation and prognostic significance. Circulation. 2001;103(17):2153-2158.
doi pubmed - Hamazaki N, Kamiya K, Matsuzawa R, Nozaki K, Ichikawa T, Tanaka S, Nakamura T, et al. Prevalence and prognosis of respiratory muscle weakness in heart failure patients with preserved ejection fraction. Respir Med. 2020;161:105834.
doi pubmed - Kera T, Kawai H, Hirano H, Kojima M, Watanabe Y, Motokawa K, Fujiwara Y, et al. Definition of respiratory sarcopenia with peak expiratory flow rate. J Am Med Dir Assoc. 2019;20(8):1021-1025.
doi pubmed - Nagano A, Wakabayashi H, Maeda K, Kokura Y, Miyazaki S, Mori T, Fujiwara D. Respiratory sarcopenia and sarcopenic respiratory disability: concepts, diagnosis, and treatment. J Nutr Health Aging. 2021;25(4):507-515.
doi pubmed pmc - Morisawa T, Kunieda Y, Koyama S, Suzuki M, Takahashi Y, Takakura T, Kikuchi Y, et al. The relationship between sarcopenia and respiratory muscle weakness in community-dwelling older adults. Int J Environ Res Public Health. 2021;18(24):13257.
doi pubmed pmc - Neder JA. Residual exertional dyspnea in cardiopulmonary disease. Ann Am Thorac Soc. 2020;17(12):1516-1525.
doi pubmed - Del Buono MG, Arena R, Borlaug BA, Carbone S, Canada JM, Kirkman DL, Garten R, et al. Exercise intolerance in patients with heart failure: JACC state-of-the-art review. J Am Coll Cardiol. 2019;73(17):2209-2225.
doi pubmed - Kelley RC, Ferreira LF. Diaphragm abnormalities in heart failure and aging: mechanisms and integration of cardiovascular and respiratory pathophysiology. Heart Fail Rev. 2017;22(2):191-207.
doi pubmed pmc - Cargnin C, Karsten M, Guaragna J, Dal Lago P. Inspiratory muscle training after heart valve replacement surgery improves inspiratory muscle strength, lung function, and functional capacity: a randomized controlled trial. J Cardiopulm Rehabil Prev. 2019;39(5):E1-E7.
doi pubmed - Stein R, Maia CP, Silveira AD, Chiappa GR, Myers J, Ribeiro JP. Inspiratory muscle strength as a determinant of functional capacity early after coronary artery bypass graft surgery. Arch Phys Med Rehabil. 2009;90(10):1685-1691.
doi pubmed - Matos-Garcia BC, Rocco IS, Maiorano LD, Peixoto TCA, Moreira RSL, Carvalho ACC, Catai AM, et al. A home-based walking program improves respiratory endurance in patients with acute myocardial infarction: a randomized controlled trial. Can J Cardiol. 2017;33(6):785-791.
doi pubmed - Smith JR, Taylor BJ. Inspiratory muscle weakness in cardiovascular diseases: Implications for cardiac rehabilitation. Prog Cardiovasc Dis. 2022;70:49-57.
doi pubmed pmc - Arai H, Satake S. English translation of the Kihon Checklist. Geriatr Gerontol Int. 2015;15(4):518-519.
doi pubmed - Satake S, Shimokata H, Senda K, Kondo I, Toba K. Validity of total Kihon checklist score for predicting the incidence of 3-year dependency and mortality in a community-dwelling older population. J Am Med Dir Assoc. 2017;18(6):552.e1-552.e6.
doi pubmed - Sato S, Miyazaki S, Tamaki A, Yoshimura Y, Arai H, Fujiwara D, Katsura H, et al. Respiratory sarcopenia: A position paper by four professional organizations. Geriatr Gerontol Int. 2023;23(1):5-15.
doi pubmed - Dall'Ago P, Chiappa GR, Guths H, Stein R, Ribeiro JP. Inspiratory muscle training in patients with heart failure and inspiratory muscle weakness: a randomized trial. J Am Coll Cardiol. 2006;47(4):757-763.
doi pubmed - Palau P, Dominguez E, Nunez E, Ramon JM, Lopez L, Melero J, Bellver A, et al. Inspiratory muscle function and exercise capacity in patients with heart failure with preserved ejection fraction. J Card Fail. 2017;23(6):480-484.
doi pubmed - Suzuki M, Teramoto S, Sudo E, Ogawa K, Namekawa T, Motrita K, Matsuse T, et al. [Age-related changes in static maximal inspiratory and expiratory pressures]. Nihon Kyobu Shikkan Gakkai Zasshi. 1997;35(12):1305-1311.
pubmed - Granger CV, Hamilton BB, Linacre JM, Heinemann AW, Wright BD. Performance profiles of the functional independence measure. Am J Phys Med Rehabil. 1993;72(2):84-89.
doi pubmed - Freiberger E, de Vreede P, Schoene D, Rydwik E, Mueller V, Frandin K, Hopman-Rock M. Performance-based physical function in older community-dwelling persons: a systematic review of instruments. Age Ageing. 2012;41(6):712-721.
doi pubmed - Guralnik JM, Simonsick EM, Ferrucci L, Glynn RJ, Berkman LF, Blazer DG, Scherr PA, et al. A short physical performance battery assessing lower extremity function: association with self-reported disability and prediction of mortality and nursing home admission. J Gerontol. 1994;49(2):M85-94.
doi pubmed - ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories. ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med. 2002;166(1):111-117.
doi pubmed - Wolstein MF, Folstein SE, McHugh PR. "Mini-mental state". A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res. 1975;12(3):189-198.
doi pubmed - Tsuchiya A, Ikeda S, Ikegami N, Nishimura S, Sakai I, Fukuda T, Hamashima C, et al. Estimating an EQ-5D population value set: the case of Japan. Health Econ. 2002;11(4):341-353.
doi pubmed - van Hout B, Janssen MF, Feng YS, Kohlmann T, Busschbach J, Golicki D, Lloyd A, et al. Interim scoring for the EQ-5D-5L: mapping the EQ-5D-5L to EQ-5D-3L value sets. Value Health. 2012;15(5):708-715.
doi pubmed - Makita S, Yasu T, Akashi YJ, Adachi H, Izawa H, Ishihara S, Iso Y, et al. JCS/JACR 2021 guideline on rehabilitation in patients with cardiovascular disease. Circ J. 2022;87(1):155-235.
doi pubmed - Fletcher GF, Ades PA, Kligfield P, Arena R, Balady GJ, Bittner VA, Coke LA, et al. Exercise standards for testing and training: a scientific statement from the American Heart Association. Circulation. 2013;128(8):873-934.
doi pubmed - Borg GA. Perceived exertion. Exerc Sport Sci Rev. 1974;2:131-153.
pubmed - Persinger R, Foster C, Gibson M, Fater DC, Porcari JP. Consistency of the talk test for exercise prescription. Med Sci Sports Exerc. 2004;36(9):1632-1636.
pubmed - Adamopoulos S, Schmid JP, Dendale P, Poerschke D, Hansen D, Dritsas A, Kouloubinis A, et al. Combined aerobic/inspiratory muscle training vs. aerobic training in patients with chronic heart failure: The Vent-HeFT trial: a European prospective multicentre randomized trial. Eur J Heart Fail. 2014;16(5):574-582.
doi pubmed - Hermes BM, Cardoso DM, Gomes TJ, Santos TD, Vicente MS, Pereira SN, Barbosa VA, et al. Short-term inspiratory muscle training potentiates the benefits of aerobic and resistance training in patients undergoing CABG in phase II cardiac rehabilitation program. Rev Bras Cir Cardiovasc. 2015;30(4):474-481.
doi pubmed pmc - Hamazaki N, Kamiya K, Yamamoto S, Nozaki K, Ichikawa T, Matsuzawa R, Tanaka S, et al. Changes in respiratory muscle strength following cardiac rehabilitation for prognosis in patients with heart failure. J Clin Med. 2020;9(4):952.
doi pubmed pmc - Winkelmann ER, Chiappa GR, Lima CO, Viecili PR, Stein R, Ribeiro JP. Addition of inspiratory muscle training to aerobic training improves cardiorespiratory responses to exercise in patients with heart failure and inspiratory muscle weakness. Am Heart J. 2009;158(5):768.e761-767.
doi pubmed - Dos Santos TD, Pereira SN, Portela LOC, Cardoso DM, Lago PD, Dos Santos Guarda N, Moresco RN, et al. Moderate-to-high intensity inspiratory muscle training improves the effects of combined training on exercise capacity in patients after coronary artery bypass graft surgery: A randomized clinical trial. Int J Cardiol. 2019;279:40-46.
doi pubmed - Miozzo AP, Stein C, Marcolino MZ, Sisto IR, Hauck M, Coronel CC, Plentz RDM. Effects of high-intensity inspiratory muscle training associated with aerobic exercise in patients undergoing CABG: randomized clinical trial. Braz J Cardiovasc Surg. 2018;33(4):376-383.
doi pubmed pmc - Soubra R, Chkeir A, Novella JL. A systematic review of thirty-one assessment tests to evaluate mobility in older adults. Biomed Res Int. 2019;2019:1354362.
doi pubmed pmc - Rinaldo L, Caligari M, Acquati C, Nicolazzi S, Paracchini G, Sardano D, Giordano A, et al. Functional capacity assessment and Minimal Clinically Important Difference in post-acute cardiac patients: the role of Short Physical Performance Battery. Eur J Prev Cardiol. 2022;29(7):1008-1014.
doi pubmed - Singh SJ, Puhan MA, Andrianopoulos V, Hernandes NA, Mitchell KE, Hill CJ, Lee AL, et al. An official systematic review of the European Respiratory Society/American Thoracic Society: measurement properties of field walking tests in chronic respiratory disease. Eur Respir J. 2014;44(6):1447-1478.
doi pubmed - Erickson KI, Voss MW, Prakash RS, Basak C, Szabo A, Chaddock L, Kim JS, et al. Exercise training increases size of hippocampus and improves memory. Proc Natl Acad Sci U S A. 2011;108(7):3017-3022.
doi pubmed pmc - Kanazawa N, Yamada S, Fushimi K. Trends in the use of cardiac rehabilitation in Japan between 2010 and 2017 - An epidemiological survey. Circ Rep. 2021;3(10):569-577.
doi pubmed pmc - Kitzman DW, Whellan DJ, Duncan P, Pastva AM, Mentz RJ, Reeves GR, Nelson MB, et al. Physical rehabilitation for older patients hospitalized for heart failure. N Engl J Med. 2021;385(3):203-216.
doi pubmed pmc
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


