| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website https://www.cardiologyres.org |
Original Article
Volume 14, Number 3, June 2023, pages 167-175
Preliminary Study on the Association of Serum Branched-Chain Amino Acids With Lipid and Hepatic Markers
Hari Krishnan Krishnamurthya, c, d , Swarnkumar Reddyb, d
, Vasanth Jayaramana, Karthik Krishnaa, Qi Songb, Tianhao Wanga, Kang Beia, John J. Rajasekarana
aVibrant Sciences LLC., San Carlos, CA 94070, USA
bVibrant America LLC., San Carlos, CA, USA
cThese authors contributed equally to this article.
dCorresponding Author: Hari Krishnan Krishnamurthy, Vibrant Sciences LLC., San Carlos, CA 94070, USA; Swarnkumar Reddy, Vibrant America LLC., San Carlos, CA, USA
Manuscript submitted November 23, 2022, accepted April 27, 2023, published online May 26, 2023
Short title: Serum BCAAs and Lipid and Hepatic Markers
doi: https://doi.org/10.14740/cr1454
| Abstract | ▴Top |
Background: Serum levels of branched-chain amino acids (BCAAs) are associated with various vital physiological functions and thus elevation in circulating levels results in several metabolic disturbances. Serum levels of BCAAs are strong predictors of various metabolic disorders. Their association with cardiovascular health is uncertain. The study aimed to investigate the association of BCAAs with circulating levels of vital cardiovascular and hepatic markers.
Methods: The study population of 714 individuals was included from the population tested for the vital cardio and hepatic biomarkers at the Vibrant America Clinical Laboratories. The subjects were stratified into four quartiles based on the serum levels of BCAAs, and their association with vital markers was studied using the Kruskal-Wallis test. Pearson’s correlation analyzed the univariant relationship of BCAAs with selected cardio and hepatic markers.
Results: BCAAs exhibited a strong negative correlation with serum HDL. Serum triglycerides were found to have a positive correlation with serum levels of leucine and valine. Univariant analysis exhibited a strong negative correlation between serum levels of BCAAs and HDL, and a positive correlation was observed between triglycerides and amino acids isoleucine and leucine. Among analyzed hepatic markers, alanine transaminase exhibited a considerable association with BCAAs.
Conclusions: The elevated levels of serum BCAAs are strongly associated with serum HDL and triglycerides. Consumption of these supplements must be in coordination with healthcare providers to avoid metabolic and cardiovascular risk.
Keywords: BCAAs; HDL; Isoleucine; Valine; Leucine; Cardiovascular disorder
| Introduction | ▴Top |
Chronic metabolic diseases such as cardiovascular diseases (CVDs), arteriosclerosis, chronic heart failure, and diabetes are the major causes of morbidity and mortality in the general population. The prevalence of cardiometabolic and CVDs has increased in the last few decades due to significant various anthropological and behavioral changes [1, 2]. Reduced physical activity, decreases in the sleep cycle, and the introduction of an energy-dense and palatable diet are the major reasons for these diseases [3]. Diabetes is the most chronic disease and is strongly associated with various cardiovascular complications which tend to increase in epidemic proportion in recent decades. Metabolic diseases are characterized by altered levels of vital metabolites [4]. For instance, reduced high-density lipoprotein (HDL) and elevated triglycerides result in the development of insulin resistance with metabolic syndrome. Elevated levels of circulating low-density lipoprotein (LDL) also represent the most vital cardiovascular risk factor. Several studies have reported the positive correlation of branched-chain amino acids (BCAAs) with various metabolic disorders including diabetes and its linked CVDs [5, 6]. Various experimental studies have been put forth to understand the underlying interrelated pathology of diabetes-linked CVDs. A study on plasma metabolite profiling using high-throughput technologies demonstrated the strong association of aromatic amino acids and BCAAs with type 2 diabetes.
BCAAs (isoleucine, leucine, and valine) are a class of essential amino acids with an aliphatic side chain and are vital for regulating various physiological functions in the human body [7]. The circulating levels of BCAA have been observed to have a consistent association with diabetes, and dysfunctional BCAA amino acids result in the development of cardiac insulin resistance, thereby the development of type 2 diabetes. Studies have proved that dysfunctions in the metabolism of BCAA result in poor cardiometabolic health, and several cross-sectional studies have proved the association of BCAA with several cardiometabolic risk factors such as insulin resistance impaired fasting glucose, elevated blood pressure, and other risk factors of coronary artery disease [5, 8, 9]. Cross-sectional studies have failed to explain the underlying mechanism of BCAA-associated CVD. To overcome this limitation, a few prospective studies have reported the role of BCAA in CVD. The elevated serum levels of BCAA result in increasing the serum levels of triglycerides. Another cross-sectional study reported that despite increasing the levels of triglycerides, BCAA also tends to decrease the circulating levels of HDL [10]. Studies have reported no correlation between serum BCAA and LDL, but an in vitro study by Halama et al [11] reported the biosynthesis of cholesterol from the byproducts of leucine. Excessive consumption of BCAA and the resulting increase in the circulating levels of BCAA may affect the transportation of tryptophan to the brain and inhibit serotonin synthesis [12]. In vivo, studies have proved the role of BCAA in the regulation of appetite and increased obesity. However, there was no clear consensus on the correlation of BCAA with vital cardiovascular markers.
We, therefore in the present study, aimed to evaluate the association of BCAA on the serum levels of various vital cardiovascular and hepatic markers. A retrospective analysis was carried out to evaluate the interrelationship of serum BCAA with lipid and hepatic markers.
| Materials and Methods | ▴Top |
Study design
This study was a retrospective study based on clinical data. The study population of 714 individuals was recruited from the population tested for the vital cardio and hepatic biomarkers at the Vibrant America Clinical Laboratories.
Ethical compliance
The study was carried out in deidentified clinical samples, hence exempted from formal ethical clearance. IRB exemption (work order #1-1098539-1) was determined by the Western Institutional Review Board (WIRB) for Vibrant America Biorepository to use delinked and deidentified remnant human specimens and medical data for research purposes. The study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration.
Determination of serum biomarkers and amino acids
Blood samples were drawn and processed for the separation of serum. Serum levels of total cholesterol were determined by an enzymatic assay catalyzed by cholesterol dehydrogenase and estimated by the Beckman Coulter AU680 analyzer. Particle enhanced immunoturbidometric assay was followed to estimate the serum levels of apolipoprotein A1 (ApoA1), Apo B, and lipoprotein (a) (Lp (a)) using a Roche Cobas 6000 C 501 analyzer. The enzyme-based colorimetric assay was used to measure the serum levels of LDL, HDL, and triglycerides via the Beckman Coulter AU680 analyzer. An electrochemiluminescence immune assay was used for the determination of N-terminal pro-B-type natriuretic peptide (NT-proBNP). The assay is based on the interaction between the antigen (NT-proBNP), a biotinylated monoclonal antibody, and a ruthenium-labeled monoclonal antibody specific to NT-proBNP. Total serum homocysteine levels were estimated by in vitro quantitative enzyme-linked immunosorbent assay using Beckman Counter AU series analyzers. The serum level of homocysteine is measured as the inverse of the amount of NADH converted to NAD + which can be measured at 340 nm. The quantitative estimation of oxidized low-density lipoprotein (oxLDL) is based on the direct sandwich technique which uses two monoclonal antibodies specific to antigenic determinants on the oxidized apolipoprotein B. The reaction is based on the reaction between the oxidized LDL in the serum with anti-oxidized LDL antibodies, the reaction is monitored spectrometrically at 450 nm. Serum myeloperoxidase was determined by a latex-enhanced immuno-turbidimetric assay based on antigen-antibody interaction. The myeloperoxidase (MPO) in the serum binds to a specific anti-MPO antibody coated on the latex. This causes agglutination and the turbidity of agglutination is directly measured as the concentration of MPO in the serum. The in vitro quantification of lipoprotein-associated phospholipase A2 (Lp-PLA2) is an enzymatic assay based on the hydrolysis of the sn-2 position of the substrate and produces a colored end product 4-nitrophenol. The change in the absorbance with the production of 4-nitrophenol is measured as the Lp-PLA2 activity.
The hepatic markers comprised vital liver enzymes such as alkaline phosphatase (ALK), aspartate transaminase (AST), alanine transaminase (ALT), and other hepatic markers such as albumin, total bilirubin, and total protein.
The presence of AST was determined by a two-step enzymatic reaction, the reaction is catalyzed by the transfer of amino group between L-aspartate and 2-oxoglutarate resulting in the formation of oxaloacetate and L-glutamate. The oxaloacetate is further oxidized into NADH in the presence of malate dehydrogenase to form NAD. The catalytic AST is given as the oxidation rate of NADH which is measured as the decrease in the absorbance.
The catalytic activity between L-alanine and 2-oxoglutarate determines the enzyme activity of ALT. L-lactate and NAD are the end products of pyruvate reduction by NADH catalyzed by lactate dehydrogenase. The catalytic activity of the ALT is directly used to quantify the rate of oxidation, and the drop in absorbance is used to measure the rate photometrically.
The ability of phosphatases to cleave p-nitrophenyl phosphate onto phosphate and p-nitrophenol in the presence of magnesium and zinc is used to quantify ALK. The rise in absorbance, which is a measure of enzyme activity, is directly proportional to the amount of p-nitrophenol emitted.
The formation of a blue-green complex between anionic bromocresol green and cationic serum albumin at an optimal pH of 4.1 allows for the measurement of albumin levels in serum. The concentration of albumin can be determined directly from the blue-green complex’s color intensity. The serum bilirubin readily solubilizes and produces a red azo dye complex with 3,5-dichlorophenyl diazonium in a colorimetric diazo technique used to measure total bilirubin. The complex’s color intensity is measured photometrically and is inversely correlated with the total bilirubin concentration. Divalent copper, which combines with the protein peptides to generate a distinctive purple biuret complex, can be used to measure the overall protein concentration. The color intensity of the complex is directly proportional to the concentration of protein.
Waters TQ-XS tandem mass spectrometer coupled with liquid chromatography mass spectrometry (LCMS) was used to determine the serum levels of BCAA.
Statistical analysis
The retrospective analysis of the clinical data was performed via Java for Windows version 1.8.161. The obtained data were stratified into four quartiles based on the cut-off points at the 25th, 50th, and 75th percentile values of serum levels of BCAA. The univariant relationship of BCAA with selected cardio and hepatic was analyzed by Pearson’s correlation with significance set at P < 0.05. GraphPad Prism version 7.00 was used to perform statistical analysis, and descriptive statistics were used to express the continuous variables (mean ± standard deviation (SD), median, minimum, and maximum)
| Results | ▴Top |
The current research aimed to study the association of BCAAs with circulating levels of vital cardiovascular and hepatic markers. The cardiovascular panel comprised all vital lipid markers such as total cholesterol, LDL, HDL, triglycerides, ApoA1, ApoB, Lp (a), NT-proBNP, small dense LDL (SDLDL), oxLDL, high-sensitivity C-reactive protein (hs-CRP), homocysteine, myeloperoxidase, and Lp-PLA2. Where, the hepatic panel is comprised of vital liver markers such as ALT, AST, total bilirubin, total protein, albumin, and ALK (Table 1).
 Click to view | Table 1. Baseline Properties of the Study |
The study comprised 714 individuals with a mean age of 48.2 ± 16.0, and the subjects were stratified into four quartiles based on the serum levels of BCAA (isoleucine: 464.2 ± 23.1, leucine: 161.3 ± 34.5 and valine: 247.9 ± 58.4) (Fig. 1). Among all the biomarkers tested the circulating levels of HDL were found to have a significant negative trend with an increase in the serum levels of all three BCAAs. ApoA1 was found to have a considerable negative trend with increasing isoleucine, whereas homocysteine showed a considerable positive trend toward increasing levels of isoleucine (Table 2). NT-proBNP and alanine transferase were also found to be weekly associated with increasing levels of serum isoleucine (Table 3). Serum HDL and ApoA1 exhibited a strong inverse trend with increasing serum leucine. Whereas other lipid markers triglycerides, SDLDL, homocysteine, and lipoprotein-associated phospholipase 2 exhibited a strong positive trend with increasing serum leucine. Among analyzed hepatic markers, ALT exhibited a strong positive trend towards an increase in serum leucine. Serum levels of valine were found to have a negative correlation with circulating HDL and ApoA1 levels. ALT exhibited a strong positive trend toward increasing serum valine (Table 4). Serum levels of homocysteine, oxLDL, Lp-PLA2, AST, and albumin were also found to be weakly associated with valine concentrations.
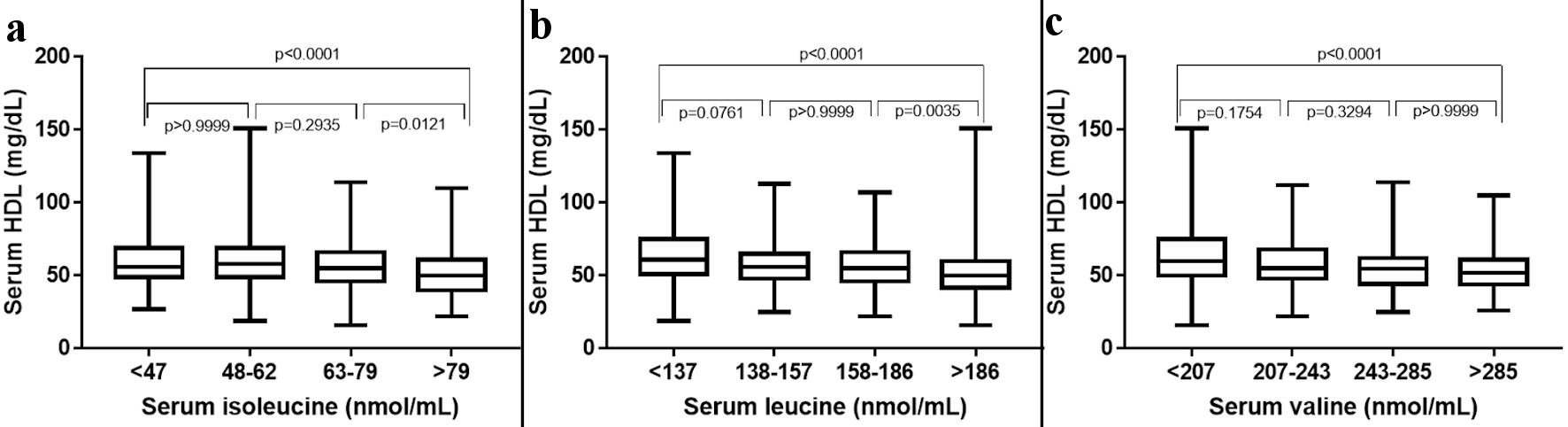 Click for large image | Figure 1. The 25th, 50th, and 75th percentiles were used as cut-off points to stratify participants according to the plasma levels of individual BCAAs. HDL: high-density lipoprotein; BCAAs: branched-chain amino acids. |
 Click to view | Table 2. Quartiles of Serum Levels of Isoleucine |
 Click to view | Table 3. Quartiles of Serum Levels of Leucine |
 Click to view | Table 4. Quartiles of Serum Levels of Valine |
Quantitative analysis by Pearson correlation exhibited the distinct correlation of the vital biomarkers with the serum levels of BCAAs. A strong negative correlation was observed between serum levels of HDL and all three BCAAs (Figs. 2-4). Where in the case of triglycerides a strong positive correlation was observed between isoleucine (r = 0.15, P < 0.0001) and leucine (r = 0.16, P < 0.0001). ApoA1 was found to have a strong negative correlation with leucine (r = -0.23, P < 0.0001) and valine (r = -0.16, P < 0.0001). Amino acids isoleucine (r = 0.10, P < 0.01) and leucine (r = 0.09, P < 0.02) were found to be positively correlated with serum homocysteine. Interestingly no significant correlation was observed with hepatic markers (Table 5). However, total bilirubin and albumin were found to exhibit a zero-correlation suggesting the rejection of the null hypothesis.
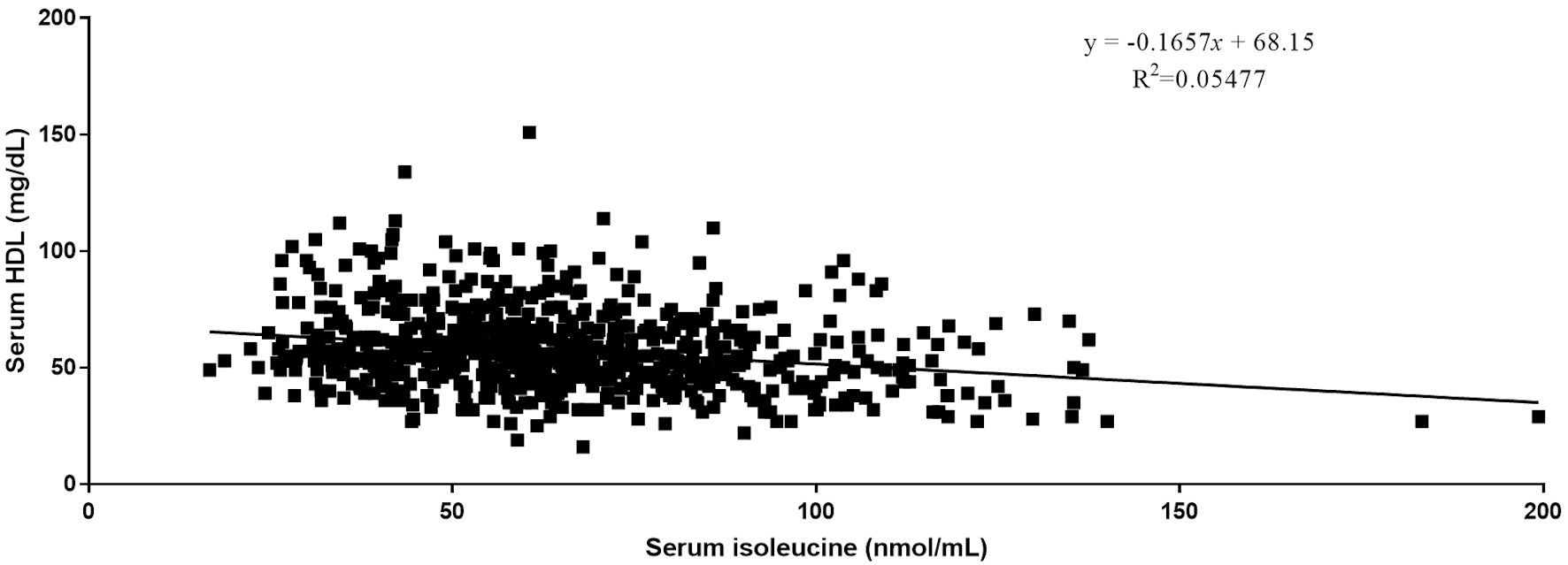 Click for large image | Figure 2. Relationship between serum isoleucine and serum HDL. HDL: high-density lipoprotein. |
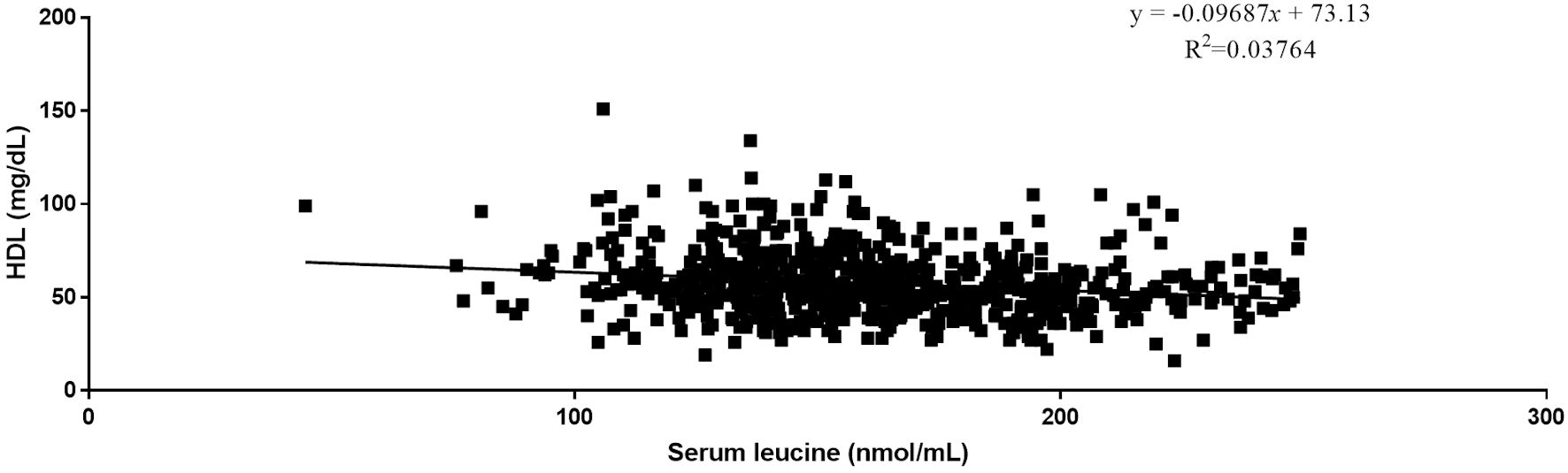 Click for large image | Figure 3. Relationship between serum leucine and serum HDL. HDL: high-density lipoprotein. |
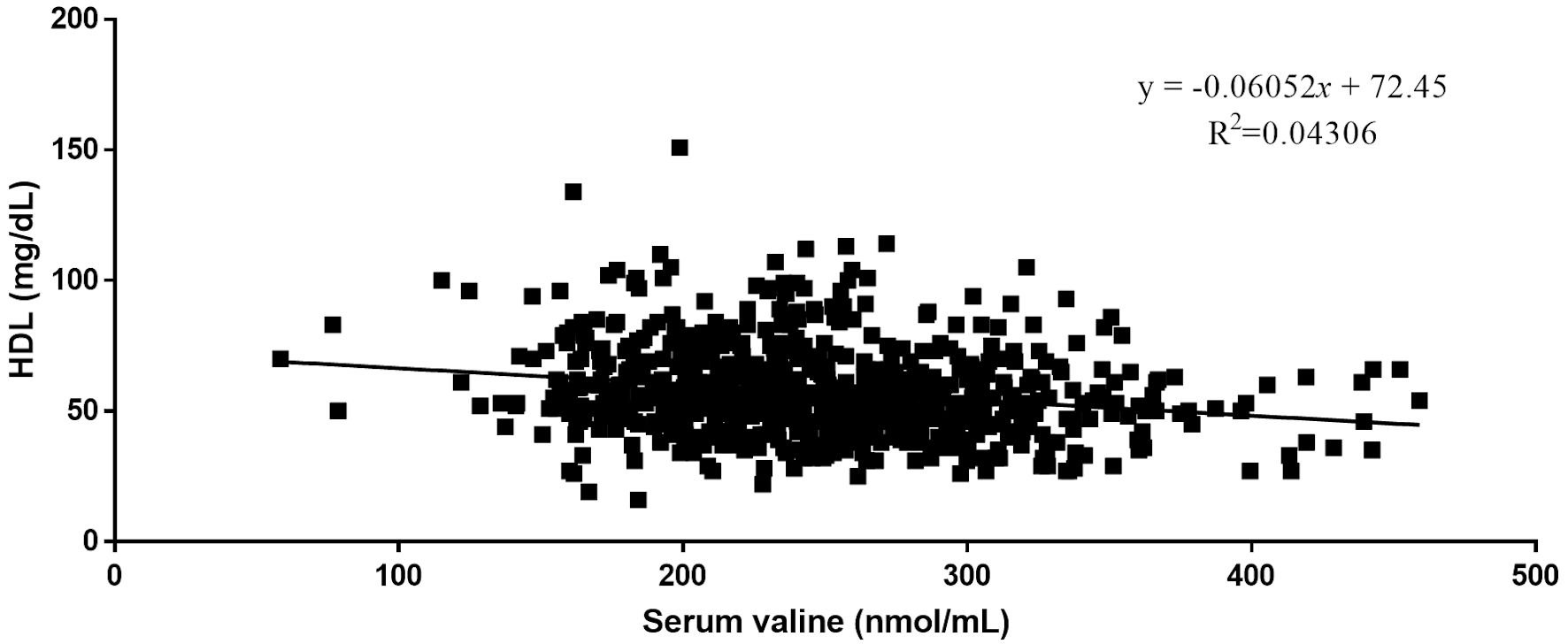 Click for large image | Figure 4. Relationship between serum valine and serum HDL. HDL: high-density lipoprotein. |
 Click to view | Table 5. Pearson Correlation of BCAA With Vital Cardio and Hepatic Biomarkers |
| Discussion | ▴Top |
The primary objective of the study is to investigate the association between circulating levels of BCAAs and vital cardio markers. The study also attempted to discover the proximity of serum BCAAs as a biomarker in the early detection of future cardiovascular events. Several metabolomic studies have reported a wide class of untargeted metabolites related to lipid metabolism, amino acid, and fatty acid metabolism associated with various CVDs. Among nine human essential amino acids, BCAAs (leucine, valine, and isoleucine) were found to have high discrimination capability in serum and various genetic and protein analyses [13]. Studies have reported that failing hearts with the accumulation of BCAAs in serum results from the downregulation of BCAA metabolism [14].
Various reports in the last decade have yielded crucial insights that are reshaping our understanding of the systems physiology of BCAA metabolism and the molecular mechanisms underlying the close relationship between BCAA homeostasis and cardiovascular health [15]. Several patient-based studies have shown that disturbances of BCAA metabolism are associated with multiple CVDs, including coronary artery disease, myocardial infarction, and heart failure [16]. However, the exact molecular mechanism remains elusive. Recent research has revealed molecular mechanisms behind BCAA catabolism in regulating cardiac metabolism and stress response [17].
BCAAs, especially leucine, are highly effective activators of mTOR signaling, which is directly implicated in cardiac hypertrophy in the heart [18]. It has been suggested that increased BCAA levels in plasma or tissue activate the mTOR pathway, although independently of Tuberous sclerosis complex (TSC) regulation. Elevated BCAA levels could lead to persistent activation of mTOR followed by serine phosphorylation of insulin receptor substrate-1 (IRS-1) via S6 kinase (p70S6K) [15]. Recent studies have suggested that leucine, a BCAA, is a key amino acid involved in mTOR activity and imparts cardioprotective effects. BCAAs selectively promote cardiac function through multiple mechanisms, including activation of mTOR [19]. In general, BCAAs play several important metabolic and physiological roles, aside from being considered as substrates for the synthesis of proteins. Reports show that BCAAs act as signaling molecules regulating the metabolism of glucose, lipid, and protein [20].
In the present study, a strong statistically significant negative correlation was observed between serum levels of BCAAs and HDL. A positive correlation was observed between triglycerides and amino acids leucine and valine. Moreover, our one-way analysis of variance (ANOVA) also proves the coexistence between the increasing levels of BCAA with serum levels of HDL. The increase in the serum levels of all three BCAAs exhibited a negative trend towards serum HDL. The current result is consistent with several previous reports. A report by Wang et al (2019) [21] indicated that elevated levels of BCAAs are directly associated with atherogenic diseases either by increasing the serum levels of triglycerides or by decreasing the HDL. They also reported that the increase in threonine may reduce the risk of atherogenic diseases. A report by Yang et al in 2016 [22] on metabolomic dyslipidemia in the Chinese Han population also reported the increase in the serum concentrations of BCAAs is associated with both elevating levels of triglycerides and decreasing the levels of HDL. High BCAAs were also associated with the development of metabolic dyslipidemia and various other metabolic disorders including diabetes [23].
Numerous studies have reported the implications of elevated levels of metabolites in the development of cardiovascular and various metabolic disorders [24]. The current study deals with the health implications of elevated levels of serum BCAAs on atherogenic lipid profiles and other related cardiovascular disorders. The molecular interactions of BCAAs with the lipid mechanism remains certainly unclear. However various in vitro studies have demonstrated that impaired BCAA catabolism results in the accumulation of BCAAs [25, 26]. Chronic levels of accumulated BCAAs result in the selective disruption of mitochondrial pyruvate utilization by inhibition of pyruvate dehydrogenase complex activity and also decreases glucose uptake, oxidation, glycogen content, and protein glycosylation. Elevated levels of BCAAs downregulate the hexosamine biosynthesis and inactivate pyruvate dehydrogenase.
BCAAs are human essential amino acids, which are the most vital among the nine essential amino acids. Overdose or elevated serum levels of these amino acids are known to be implicated in the development of CVDs. It is also proven that BCAAs or their related metabolites are proven to be independently associated with severe coronary artery disease. Several reports including the current study have reported the significant associations between BCAAs with triglycerides and HDL. Reports have been limited in the case of BCAAs and their association with hepatic markers. Adipose tissue being the main site for the interaction of BCAAs with lipid metabolites, whereas the liver seems to be another candidate organ for amino acid metabolism [27]. BCAAs are oxidized extensively in extra-hepatic tissue and are known to be correlated with non-alcoholic fatty liver disease [28]. This may result in morbidity of insulin resistance and the development of type 2 diabetes mellitus. BCAAs may also increase insulin resistance in hepatic cells. As a result of increased gluconeogenesis, altered hepatic glycogen export affects hepatic lipid homeostasis and results in the accumulation and deposit of triglycerides and other fatty acids. Although the significance of such a metabolic pattern has not been completely understood. The heart expresses the highest levels of enzymes in the BCAA catabolic pathway, but BCAA oxidation contributes to a very small fraction of energy production in the heart [25].
The study has a few limitations, such as the limited size of the study population. Additionally, since it’s a retrospective study possibility of multifactor adjustment of analysis is limited. However, our study has detailed the significant associations between BCAAs and various lipid markers along with vital hepatic markers. The wide use of BCAAs to improve the clinical indication of malnutrition has increased in recent years. This makes the need to monitor the serum levels of BCAAs important. To best of our knowledge, this is the first report to discuss the associations between lipid and hepatic markers with BCAAs.
Conclusions
In conclusion, our study demonstrates that an increase in individual or total serum BCAAs is inversely associated with HDL. Triglycerides, ApoA1, and alanine transferase exhibited a considerable association with serum BCAAs. These results present a potential rationale for the use of serum BCAAs as an effective biomarker for the diagnosis of various metabolic and cardiovascular disorders. Since BCAAs seem to modulate lipid and hepatic markers and are widely supplemented in the fitness industry further research is warranted to limit undesirable effects.
Acknowledgments
We acknowledge Vibrant America LLC for supporting this research.
Financial Disclosure
None to declare.
Conflict of Interest
Krishnamurthy, Jayaraman, Krishna, Wang, Bei, and Rajasekaran are employees of Vibrant Sciences LLC. Reddy, Song, and Rajasekaran are employees of Vibrant America LLC
Informed Consent
Not applicable
Author Contributions
Hari Krishnan Krishnamurthy, Karthik Krishna, and Tianhao Wang performed the research. Hari Krishnan Krishnamurthy, John J. Rajasekaran, and Vasanth Jayaraman designed the study. Qi Song, Kang Bei, and Swarnkumar Reddy analyzed the data. Hari Krishnan Krishnamurthy and Swarnkumar Reddy wrote the article.
Data Availability
The data used to support the findings of this study are available from the corresponding author upon request.
| References | ▴Top |
- Low Wang CC, Hess CN, Hiatt WR, Goldfine AB. Clinical update: cardiovascular disease in diabetes mellitus: atherosclerotic cardiovascular disease and heart failure in type 2 diabetes mellitus - mechanisms, management, and clinical considerations. Circulation. 2016;133(24):2459-2502.
doi pubmed pmc - Li JJ, Liu HH, Li S. Landscape of cardiometabolic risk factors in Chinese population: a narrative review. Cardiovasc Diabetol. 2022;21(1):113.
doi pubmed pmc - Astrup A. Healthy lifestyles in Europe: prevention of obesity and type II diabetes by diet and physical activity. Public Health Nutr. 2001;4(2B):499-515.
doi pubmed - Kengne AP, Amoah AG, Mbanya JC. Cardiovascular complications of diabetes mellitus in sub-Saharan Africa. Circulation. 2005;112(23):3592-3601.
doi pubmed - Hu W, Sun L, Gong Y, Zhou Y, Yang P, Ye Z, Fu J, et al. Relationship between branched-chain amino acids, metabolic syndrome, and cardiovascular risk profile in a Chinese population: a cross-sectional study. Int J Endocrinol. 2016;2016:8173905.
doi pubmed pmc - Tobias DK, Lawler PR, Harada PH, Demler OV, Ridker PM, Manson JE, Cheng S, et al. Circulating branched-chain amino acids and incident cardiovascular disease in a prospective cohort of US women. Circ Genom Precis Med. 2018;11(4):e002157.
doi pubmed pmc - Dullius A, Fassina P, Giroldi M, Goettert MI, Volken de Souza CF. A biotechnological approach for the production of branched chain amino acid containing bioactive peptides to improve human health: A review. Food Res Int. 2020;131:109002.
doi pubmed - Hamaya R, Mora S, Lawler PR, Cook NR, Ridker PM, Buring JE, Lee IM, et al. Association of plasma branched-chain amino acid with biomarkers of inflammation and lipid metabolism in women. Circ Genom Precis Med. 2021;14(4):e003330.
doi pubmed pmc - Jennings A, MacGregor A, Pallister T, Spector T, Cassidy A. Associations between branched chain amino acid intake and biomarkers of adiposity and cardiometabolic health independent of genetic factors: A twin study. Int J Cardiol. 2016;223:992-998.
doi pubmed pmc - Lent-Schochet D, McLaughlin M, Ramakrishnan N, Jialal I. Exploratory metabolomics of metabolic syndrome: A status report. World J Diabetes. 2019;10(1):23-36.
doi pubmed pmc - Halama A, Horsch M, Kastenmuller G, Moller G, Kumar P, Prehn C, Laumen H, et al. Metabolic switch during adipogenesis: From branched chain amino acid catabolism to lipid synthesis. Arch Biochem Biophys. 2016;589:93-107.
doi pubmed - van Hall G, Raaymakers JS, Saris WH, Wagenmakers AJ. Ingestion of branched-chain amino acids and tryptophan during sustained exercise in man: failure to affect performance. J Physiol. 1995;486(Pt 3):789-794.
doi pubmed pmc - Li R, Li F, Feng Q, Liu Z, Jie Z, Wen B, Xu X, et al. An LC-MS based untargeted metabolomics study identified novel biomarkers for coronary heart disease. Mol Biosyst. 2016;12(11):3425-3434.
doi pubmed - Gojda J, Cahova M. Gut microbiota as the link between elevated BCAA serum levels and insulin resistance. Biomolecules. 2021;11(10):1414.
doi pubmed pmc - McGarrah RW, White PJ. Branched-chain amino acids in cardiovascular disease. Nat Rev Cardiol. 2023;20(2):77-89.
doi pubmed - Du X, You H, Li Y, Wang Y, Hui P, Qiao B, Lu J, et al. Relationships between circulating branched chain amino acid concentrations and risk of adverse cardiovascular events in patients with STEMI treated with PCI. Sci Rep. 2018;8(1):15809.
doi pubmed pmc - Sun H, Wang Y. Branched chain amino acid metabolic reprogramming in heart failure. Biochim Biophys Acta. 2016;1862(12):2270-2275.
doi pubmed - Huang Y, Zhou M, Sun H, Wang Y. Branched-chain amino acid metabolism in heart disease: an epiphenomenon or a real culprit? Cardiovasc Res. 2011;90(2):220-223.
doi pubmed pmc - Vanweert F, Schrauwen P, Phielix E. Role of branched-chain amino acid metabolism in the pathogenesis of obesity and type 2 diabetes-related metabolic disturbances BCAA metabolism in type 2 diabetes. Nutr Diabetes. 2022;12(1):35.
doi pubmed pmc - Latimer MN, Sonkar R, Mia S, Frayne IR, Carter KJ, Johnson CA, Rana S, et al. Branched chain amino acids selectively promote cardiac growth at the end of the awake period. J Mol Cell Cardiol. 2021;157:31-44.
doi pubmed pmc - Wang FH, Liu J, Deng QJ, Qi Y, Wang M, Wang Y, Zhang XG, et al. Association between plasma essential amino acids and atherogenic lipid profile in a Chinese population: A cross-sectional study. Atherosclerosis. 2019;286:7-13.
doi pubmed - Yang P, Hu W, Fu Z, Sun L, Zhou Y, Gong Y, Yang T, et al. The positive association of branched-chain amino acids and metabolic dyslipidemia in Chinese Han population. Lipids Health Dis. 2016;15:120.
doi pubmed pmc - Yamakado M, Nagao K, Imaizumi A, Tani M, Toda A, Tanaka T, Jinzu H, et al. Plasma free amino acid profiles predict four-year risk of developing diabetes, metabolic syndrome, dyslipidemia, and hypertension in Japanese population. Sci Rep. 2015;5:11918.
doi pubmed pmc - Rask-Madsen C, Kahn CR. Tissue-specific insulin signaling, metabolic syndrome, and cardiovascular disease. Arterioscler Thromb Vasc Biol. 2012;32(9):2052-2059.
doi pubmed pmc - Li T, Zhang Z, Kolwicz SC, Jr., Abell L, Roe ND, Kim M, Zhou B, et al. Defective branched-chain amino acid catabolism disrupts glucose metabolism and sensitizes the heart to ischemia-reperfusion injury. Cell Metab. 2017;25(2):374-385.
doi pubmed pmc - Yoon MS. The emerging role of branched-chain amino acids in insulin resistance and metabolism. Nutrients. 2016;8(7):405.
doi pubmed pmc - She P, Van Horn C, Reid T, Hutson SM, Cooney RN, Lynch CJ. Obesity-related elevations in plasma leucine are associated with alterations in enzymes involved in branched-chain amino acid metabolism. Am J Physiol Endocrinol Metab. 2007;293(6):E1552-1563.
doi pubmed pmc - Della Torre S. Non-alcoholic fatty liver disease as a canonical example of metabolic inflammatory-based liver disease showing a sex-specific prevalence: relevance of estrogen signaling. Front Endocrinol (Lausanne). 2020;11:572490.
doi pubmed pmc
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


