| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website http://www.cardiologyres.org |
Original Article
Volume 10, Number 4, August 2019, pages 223-229
Secondary Prevention of Nonvalvular Atrial Fibrillation: A Retrospective Cohort Study
Renato De Vecchisa, d, Andrea Pacconeb, Marco Di Maioc
aPreventive Cardiology and Rehabilitation Unit, DSB 29 “S. Gennaro dei Poveri Hospital”, via S. Gennaro dei Poveri 25, 80136 Naples, Italy
bDepartment of Cardiology, University of Bari “Aldo Moro”, Bari, Italy
cDepartment of Cardiology, University of Campania “Luigi Vanvitelli”, 80138 Naples, Italy
dCorresponding Author: Renato De Vecchis, Preventive Cardiology and Rehabilitation Unit, DSB 29 “S. Gennaro dei Poveri Hospital”, via S.Gennaro dei Poveri 25, 80136 Naples, Italy
Manuscript submitted July 11, 2019, accepted July 23, 2019
Short title: Amiodarone Is Efficacious in Preventing AF Relapses
doi: https://doi.org/10.14740/cr909
| Abstract | ▴Top |
Background: Secondary prevention of atrial fibrillation (AF) could be carried out by means of antiarrhythmic drugs; however this strategy has not received any endorsement because these drugs are burdened by a high risk of proarrhythmic events (flecainide, sotalol) or extracardiac effects (amiodarone).
Methods: In our retrospective cohort study we have compared amiodarone 200 mg per day with the strategy implying the renunciation of any specific drug as well as with the approach using oral anticoagulant (rivaroxaban) or a combined approach including amiodarone plus rivaroxaban.
Results: A total of 255 patients with a history of AF (paroxysmal, persistent or long-lasting persistent) successfully treated with achievement of sinus rhythm have been gathered. Amiodarone has been the most effective option for AF secondary prevention, with regard to the recurrences of AF as well as rehospitalizations: P (Kruskal-Wallis test) < 0.05 for both, over a median follow-up of 24 months.
Conclusions: Patients kept free from any specific drug therapy have been shown to experience more numerous AF relapses and related rehospitalizations. On the contrary, the amiodarone use has been associated with a decreased risk of AF recurrences and hospital admissions. Thus, amiodarone might be an efficacious tool for realizing a successful long-term AF secondary prevention.
Keywords: Atrial fibrillation; Secondary prevention; Amiodarone
| Introduction | ▴Top |
Secondary prevention of nonvalvular atrial fibrillation (AF) could be implemented as necessary measure aimed to minimize the risk of AF relapses. Nevertheless, no well-defined instructions in this regard are reported in the current guidelines of the European Society of Cardiology (ESC), perhaps because all the antiarrhythmic agents suitable for this indication have the drawbacks of an incumbent risk of proarrhythmic events (flecainide, propafenone and sotalol) [1] or extracardiac effects (amiodarone) [2, 3]. Furthermore, for some antiarrhythmic drug, an increase in mortality has been demonstrated, such as quinidine [4], dronedarone when given to patients with chronic heart failure [5], and flecainide when administered to patients with significant ischemic heart disease or heart failure [6].
Moreover, secondary AF prevention with the use of drugs or of transcatheter ablation represents a well-encoded measure mainly for patients prone to frequent AF relapses rather than for patients with history of an isolate episode of AF. In addition, according to the opinion of some scholars [7-9] in several clinical settings the simply adoption of behavioral healthy habits or the appropriate therapeutic management of underlying disease such as hypertension, diabetes or chronic heart failure would be able in itself to prevent the relapses of nonvalvular AF, so as to make superfluous to resort to a specific drug prevention for hindering a possible AF recurrence.
In any case, the discussion of how to improve the patient’s prognosis after restoring sinus rhythm subsequently to an episode of nonvalvular AF is made quite complex by the fact that there is a rather wide variety of therapeutic options available for AF secondary prevention.
| Methods | ▴Top |
In the present retrospective cohort study, we investigated some endpoints concerning secondary prevention of nonvalvular AF in a number of patients all having a history of at least one episode of nonvalvular AF successfully reversed by drug therapy, by external transthoracic electric shock or by transcatheter ablation. For this study the Institutional Review Board Approval was not required, considering the study features (retrospective cohort study). Moreover, this study was conducted in compliance with the ethical standards of the responsible institution on human subjects as well as with the Helsinki Declaration. This study did not involve experiments on animals.
The endpoints of our retrospective cohort study were AF relapses, stroke or transient ischemic attacks (TIAs), all-cause death and hospital readmissions.
One of the main tasks of our team was the collection of data derived from a median follow-up of 24 months concerning the four above-mentioned endpoints. It is important to note that the pharmacological options described in the study were carried out by medical/health professionals in the field of clinical and interventional arrhythmology who do not coincide with the doctors (the article’s authors) who at a later stage extracted the archived data and performed statistical comparisons. In other words, the various treatments, with the choice of drugs and their respective dosages, were prescribed by physicians who subsequently had no role in drafting the article nor making comparative evaluations between groups and in processing and interpretation of the data.
As regards the origin of the data used for making comparisons, it is largely composite due to the fact that the pertaining database collects contributions attributable to patients belonging to several hospital institutions: the Cardiology Outpatient Units and Inpatient Divisions of the ASL Napoli 1 Centro (Naples, Italy), as well as the Arrhythmology Operational Unit of the “Pineta Grande” Clinic of Castelvolturno (CE), Italy, and the Clinical Arrhythmology and Electrophysiology Unity of the “San Raffaele” IRCCS Hospital in Milan, Italy. Four main solutions were identified for realizing a therapeutic plan against the AF relapses.
A first option was the complete renunciation of any specific drug-based prevention. In other terms, the prophylaxis was simply accomplished by avoiding any harmful behavior (e.g., strenuous efforts on the course of sport activities) or dangerous substance (caffeine, tobacco, etc.) able to elicit an AF relapse in predisposed subjects. Moreover, in this subset of patients, a great importance was given to the scrupulous adhesion to any preexisting therapy aimed to counter a possible underlying disease (ischemic or hypertensive cardiopathy, dilated or hypertrophic cardiomyopathy, diabetes, hypercholesterolemia, etc.), whose incorrect management would have favored or caused the occurrence of an AF relapse. A second option was the adoption of a drug strategy consisting of amiodarone taken orally at the dose of 200 mg per day. A third option was based on the use of an anticoagulant agent, namely rivaroxaban at the dose of 20 mg per day. The option in favor of an anticoagulant-based prevention was driven by the consideration that the administration of an anticoagulant, albeit not able to prevent a rhythm disorder such as AF, is however consistent with the purpose of efficaciously antagonizing the deleterious thromboembolic consequences of AF, namely the embolic events that originate from the relatively rapid formation (even after only 48 h) of thrombotic masses within the left atrial appendage, with subsequent dangerous dissemination of embolic fragments in the bloodstream with the potential for peripheral or splanchnic or cerebral emboli. A fourth choice was the combined antiarrhythmic and antithrombotic prophylaxis realized with a drug regimen of amiodarone (200 mg once daily) plus rivaroxaban (20 mg once daily). Importantly, the physicians prescribing operational strategies for secondary prevention of AF (nothing, amiodarone or rivaroxaban or both) almost always omitted to specify in the clinical sheet the guiding criteria and/or rational motivations for the strategy to be applied to individual patients. In fact, from the thorough analysis of the clinical picture (in particular, possible comorbidities, possible coexistence of signs and symptoms of heart failure, and echocardiographic measurements of the left atrium), as well as from the chronological characterization of the previous AF (whether paroxysmal, persistent, or long-lasting persistent), it was not consistently possible to identify a rational criterion, capable of systematically justifying the doctor’s choice to avoid any specific preventive measure or to use the antiarrhythmic, anticoagulant or combined strategy for AF secondary prevention. The retrospective review of the medical records that were used in the study preparation involved the years 2017 and 2016, and 2015 in part. In any case these time limits allowed the authors to collect a sufficient number of patients to be included in the statistical comparisons necessary to the retrospective study and to the article’s preparation. Moreover, patients were informed about their inclusion in anonymous form in the analysis of aggregate data and they consistently consented being included in this retrospective study, after being made aware of its modalities and scientific aims.
Statistical analysis
Continuous data were reported as means (± standard deviation (SD)) or medians (interquartile range (IQR)). Kolmogorov-Smirnov test was used to check for normality. Comparisons between groups were performed using the Student’s t-test or Mann-Whitney U test where appropriate. Categorical data were reported as proportions or percentages and comparisons between groups were performed using the Chi-squared test or the Fisher’s exact test where appropriate. Furthermore, Kruskal-Wallis test was carried out for making comparisons. Contingency tables using Chi-squared test were also employed, with the four modalities of AF secondary prevention (no specific medication, amiodarone, rivaroxaban, combined regimen with amiodarone and rivaroxaban) being assumed as exposure variables within four separate tables, each applied to one of the four outcome variables investigated in the study (AF relapses , stroke or TIA, all-cause death and hospital readmissions). Statistical tests were two-tailed, and a P value < 0.05 was considered statistically significant. Analyses were conducted using Excel 2016 (version 16.0, Seattle, WA, USA) as well as MedCalc Version 18.6 (Acacialaan 22, 8400 Ostend, Belgium) and Epi-Info version 7.1.5.0 for Windows (Centers for Disease Control and Prevention, Atlanta, Georgia (USA).
| Results | ▴Top |
In our retrospective investigation, the overall study population consisted of 255 patients, all of whom had experienced at least one episode of nonvalvular AF followed by successful cardioversion. The risk of developing an AF recurrence was explored through the whole range of the secondary prevention approaches, namely no pharmacological prevention, antiarrhythmic prevention by means of oral amiodarone (200 mg per day), anticoagulant prevention by means of oral rivaroxaban (20 mg per day) and combined, antiarrhythmic and anticoagulant prevention, with amiodarone plus rivaroxaban(Figs. 1-4). The median follow-up of this retrospective study was 24 months (interquartile range: 18 - 28 months).
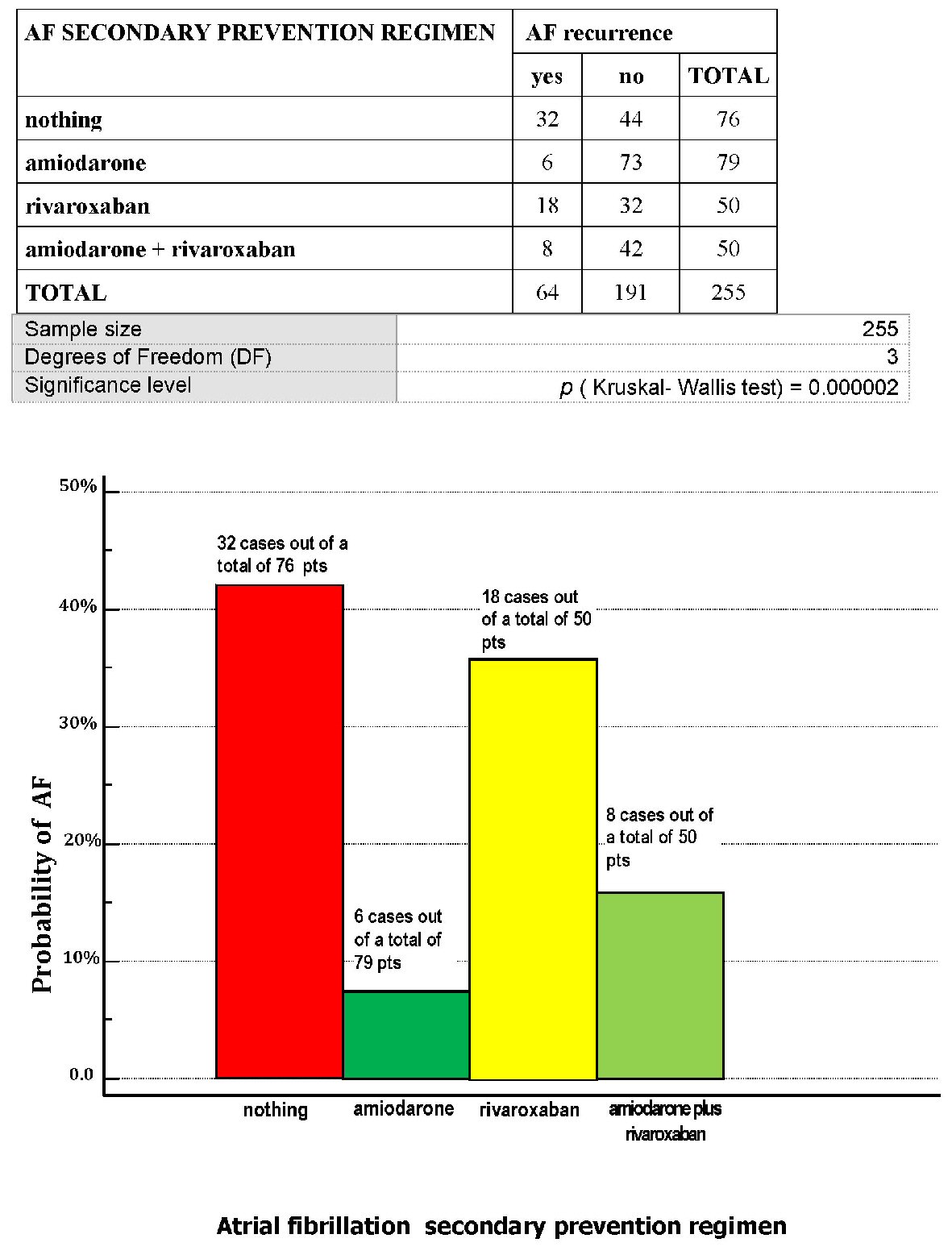 Click for large image | Figure 1. Through a series of patients retrospectively gathered, who had been involved in a program of AF secondary prevention, the distribution of the probability of atrial fibrillation (AF) relapse has been searched for, depending on the type of approach established for every patient: no drugs (“nothing”), amiodarone 200 mg per day, rivaroxaban 20 mg per day, amiodarone 200 mg per day plus rivaroxaban 20 mg per day. AF: atrial fibrillation; pts: patients. |
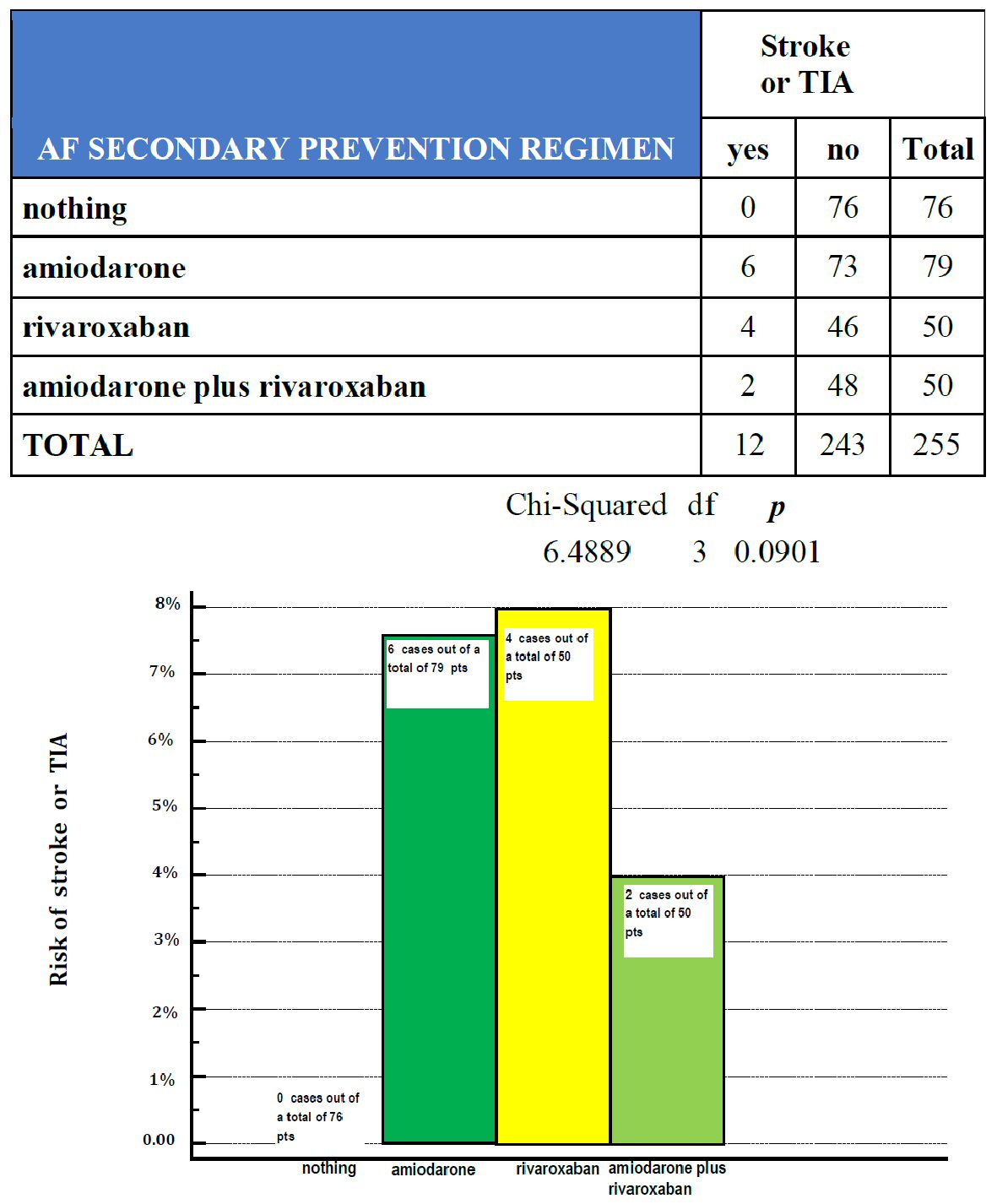 Click for large image | Figure 2. Across a series of patients with a history of AF, retrospectively gathered according to a design of retrospective cohort study, the respective probability of being involved by a stroke or TIA has been investigated for each of the above-reported cohorts: patients who had been exempted from any specific drug (nothing, 76 patients), patients taking amiodarone 200 mg per day (amiodarone, 79 patients), patients subjected to prophylaxis with rivaroxaban 20 my per day (rivaroxaban, 50 patients) and patients taking both amiodarone 200 mg per day and rivaroxaban 20 mg per day (amiodarone plus rivaroxaban, 50 patients). TIA: transient ischemic attacks; pts: patients; df: degrees of freedom. |
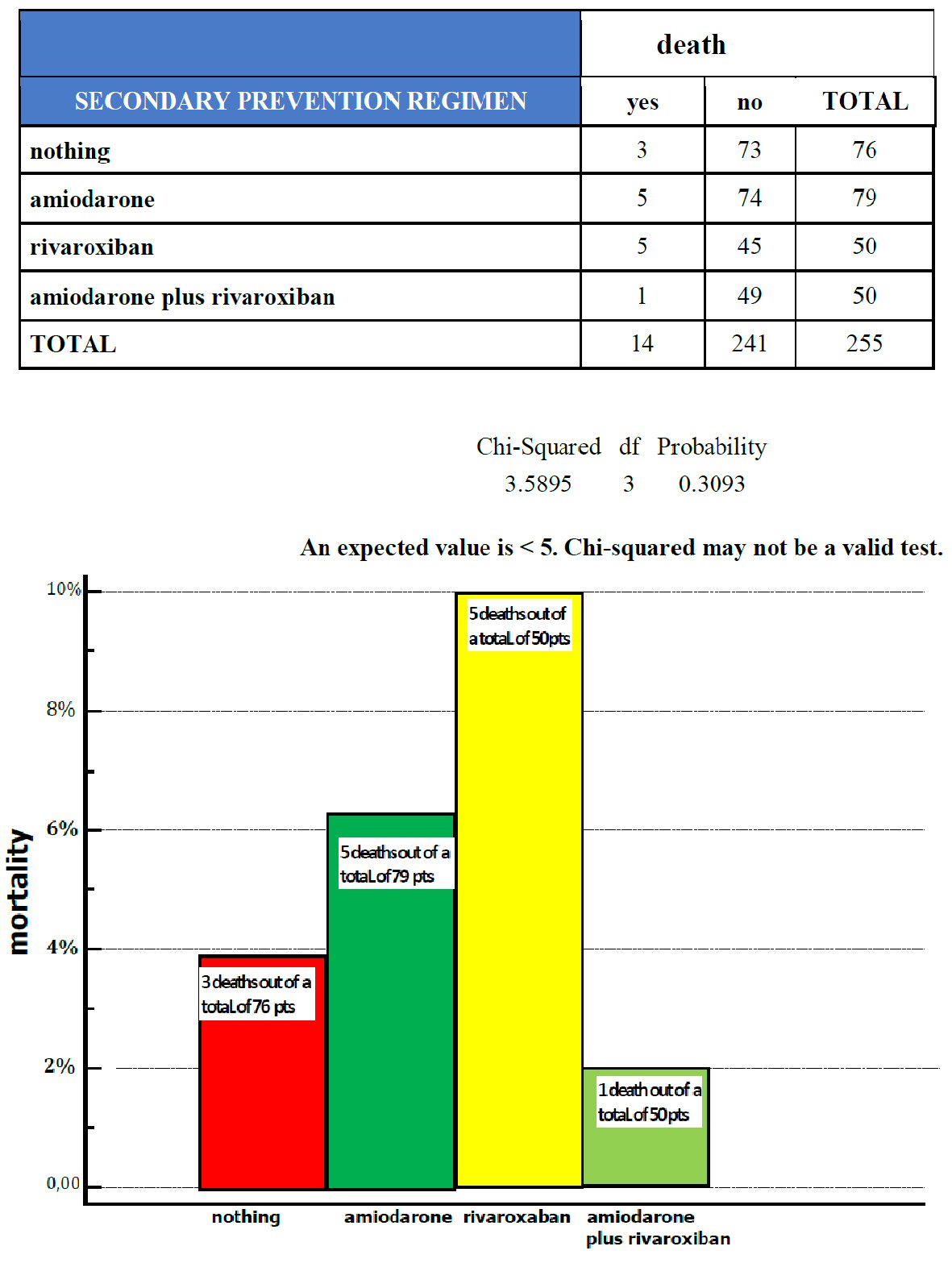 Click for large image | Figure 3. The distribution of deaths is represented within the four cohorts of patients with a history of atrial fibrillation (AF), subjected to different regimens of AF secondary prevention: no drugs (nothing, 76 patients), amiodarone (79 patients), rivaroxaban (50 patients) and combined administration of amiodarone plus rivaroxaban (50 patients). Legend: pts, patients. |
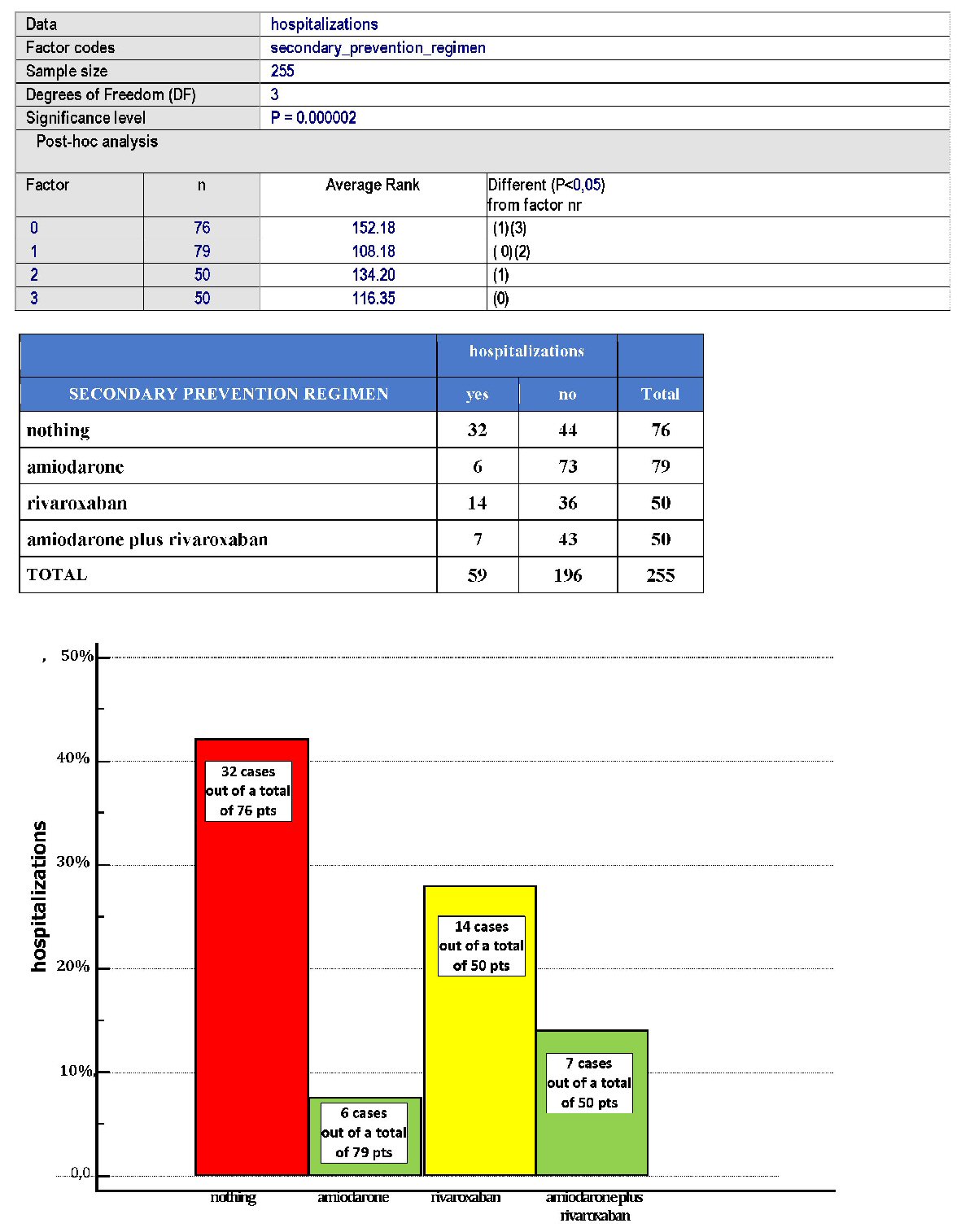 Click for large image | Figure 4. Distribution of hospitalizations within the four cohorts of patients with a history of AF. |
The patients undergoing secondary AF prevention with one of the four abovementioned measures had had their previous cardioversion realized by means of various interruption modalities such as oral or intravenous pharmacologic conversion to sinus rhythm, external transthoracic electric shock and radiofrequency transcatheter ablation. Fifty-five out of a total of 255 patients (21.5%) had a history of more than one episode of nonvalvular AF, and in almost all the cases AF had showed the characteristics of paroxysmal arrhythmia. Whatever treatment was applied to the first episode of AF (pharmacological or electrical cardioversion, or transcatheter ablation), patients were usually discharged the day after the achievement of sinus rhythm, and oral anticoagulation was continued for at least 3 months.
The percentage distribution of the various ways of pharmacologic cardioversion was as follows, intravenous amiodarone: 115 patients; intravenous propafenone: 70 patients; flecainide per os: 35 patients; hydroquinidine plus digoxin: 35 patients. In addition to these pharmacotherapeutic modalities, other non-drug-based modalities were also implemented, including: external transthoracic electric shock: 15 cases; trans-catheter ablation (Abl): 12 cases. Abl was always carried out not as a way of preservation of the previously achieved sinus rhythm, but as an intervention to restore sinus rhythm.
Judging from the medical records gathered in the course of retrospective investigation, we found that drug therapy was adopted as a first choice treatment in 100% of cases. In fact, external transthoracic electric shock or Abl were always accomplished due to the failure of the pharmacological approach, including the cases of arrhythmia resistant to pharmacological cardioversion, as well as the cases in which pharmacological cardioversion had been achieved, but it had been ephemeral because an almost immediate relapse had occurred a few hours or days after the transient success of the pharmacological approach. In Figures 1-4 contingency tables offer for the reader a synopsis of the effects exerted by the four secondary AF prevention strategies on each of the four endpoints investigated in the retrospective study: AF relapses, cerebral thromboembolic events, all-cause death, and rehospitalizations.
Importantly, amiodarone was the most effective method of secondary prevention, with regard to the recurrences of AF or rehospitalizations, P (Kruskal-Wallis test) < 0.05 for both (Figs. 1 and 4).
In our retrospective research we then found that 76 out of a total of 255 patients (equal to 29.8%) did not adopt any specific antiarrhythmic measure against AF nor any anticoagulant-based anti-embolic protection. Nevertheless this subset of patients did not show any documented episode of cardioembolic event (Fig. 2). By contrast, as many as 32 AF episodes were recorded within this subset (Fig. 1). A possible reason for this discrepancy, i.e., the ascertained relatively high incidence of AF episodes conflicting with lack of cardioembolic strokes or TIAs is reported in the Discussion.
Among the various types of anticoagulants available for long-term antithrombotic prophylaxis of patients with history of nonvalvular AF, our research considered only patients treated with rivaroxaban, in order to correspond to criteria of homogeneity and standardized prevention strategy. Thus, patients receiving another type of oral anticoagulant were excluded from the study. In rivaroxaban group, four out of a total of 50 patients (8%) were shown to be involved by newly diagnosed cerebral events (Fig. 2). For them the analysis of medical records did not allow to detect with certainty a cardioembolic origin in two out of four cases.
Side effects were reported for amiodarone as well as for rivaroxaban: a moderate dysthyroidism without clinical signs or symptoms was found in 32 out of a total of 129 patients taking amiodarone (24.8%), while among patients taking rivaroxaban (100 on the whole) the side effects were hematuria (nine patients), epistaxis (eight patients), minor gastroenteric bleeding (seven patients).
| Discussion | ▴Top |
Across a series of 255 patients retrospectively grouped according to the modality adopted for secondary prevention of nonvalvular AF (no specific drug-based prevention, amiodarone 200 mg per day, rivaroxaban 20 mg per day, combined regimen amiodarone plus rivaroxaban), amiodarone at the dose of 200 mg per day yielded a significantly superior protection against AF relapses or rehospitalizations. Contrariwise, the choice to avoid any specific drug in patients who had experienced nonvalvular AF was associated with an increased risk of AF relapses and rehospitalization over a median follow-up of 24 months.
It is appropriate to remember here that no specific long-term antiarrhythmic prophylaxis has been established until now in AF patients following their conversion to sinus rhythm [10] by means of pharmacological or electrical cardioversion. Instead, only in the case of ablated patients, during the so-called blanking period, that is the time frame of 3 months following the ablative procedure, the indication for an antiarrhythmic and anticoagulant treatment is clearly codified as an imperative and inescapable rule [11]. Therefore our research deals with a topic that is still largely available for scholarly interpretations because it is not still subject to cogent and well codified norms. In practice, secondary prevention of AF recurrences might or might not be implemented, at complete discretion of the treating physician. In fact, no binding guidelines exist on this point. In addition, based on the comparison of Figure 1with Figure 2, a discrepancy somewhat would exist between the high incidence of AF relapses and the lack of cardioembolic events, both noticeable in the patient group not undergoing any specific drug treatment. An explanation for this may be the latency time, usually not shorter than 48 h, which separates the onset of AF from the formation of thrombi within the left atrial appendage. Thus, when timely started, an anticoagulant treatment in addition to the antiarrhythmic therapy is likely to prevent the formation of thrombi during an episode of AF. In this regard, it is appropriate to remind here that in the hospital institutions from which the case record was derived, on the occasion of any recurrence of AF, patients were treated with prompt administration of intravenous unfractionated heparin which was progressively integrated with warfarin or with a new oral anticoagulant (usually rivaroxaban) until to complete heparin replacement by anticoagulant. Thus, the timeliness and appropriateness of the anticoagulant treatment might explain the finding of a discrepancy between a relatively high number of AF relapses on the one hand, and the lack of cardioembolic events, on the other. Furthermore, we would like to disapprove here the use of anticoagulants for purposes of secondary prevention of AF. In fact, it should rather be conducted with an electrophysiologic agent, such as amiodarone or a different antiarrhythmic drug, in the case of proven failure of this agent in preventing AF relapses in a given patient.
Secondary prophylaxis with amiodarone is likely to be less risky and more rational compared to that achieved with an anticoagulant. Treating physicians should be aware of the side effects, stemming from the long-term use of an anticoagulant e.g., gastroenteric minor bleedings causing debilitating anemia sometimes a little bit sneaky or underrated, compared to the risks related to the amiodarone use. The latter, in our opinion, especially in patients with left atriomegaly with a higher risk of AF recurrence, might be the first-choice drug for secondary prevention of AF. However, this indication is not codified by the current ESC guidelines [10] mainly due to the fact that chronic amiodarone administration harms the thyroid, causing a reversible iatrogenic hypothyroidism; while the amiodarone-related hyperthyroidism, albeit possible, is rare. In truth, the indications provided by the ESC guidelines on the issue of AF prevention with the use of antiarrhythmics definitely diverge from our inferences, considering that they make a favorable judgment (1A) exclusively on amiodarone applied to AF prevention in chronic heart failure patients. On the contrary, in the current ESC guidelines [10] the overall evaluation of this molecule for purposes of secondary AF prevention leads to the statement that “amiodarone is a second-choice therapy in many patients because of its extracardiac side effects”. Furthermore, amiodarone would be ineffective in preventing AF relapses when tested in ablated patients immediately after the “blanking period” [12].
In any case, based on our albeit limited study population (255 patients), amiodarone has been shown to be superior to both the strategy implying the intentional renunciation of any specific drug and the anticoagulant strategy entailing the chronic use of non-vitamin K antagonist oral anticoagulant.
Conclusions
Based on a study population of 255 patients with history of nonvalvular AF, our retrospective cohort study has evidenced that long-term oral amiodarone administration (200 mg per day) can serve as an efficacious measure able to preserve patients from AF relapses and related hospitalizations. Amiodarone has been shown to be superior to the other tested strategies, i.e., no drugs, as well as rivaroxaban or combined regimen amiodarone plus rivaroxaban, for purposes of secondary AF prevention.
Acknowledgments
None to declare.
Financial Disclosure
The authors Renato De Vecchis, Andrea Paccone and Marco Di Maio declare that the present article has not benefitted from any source of funding.
Conflict of Interest
The authors Renato De Vecchis, Andrea Paccone and Marco Di Maio do not have any conflicts of interest to declare concerning the present article.
Informed Consent
Not applicable.
Author Contributions
RDV and AP contributed to conceptualization; RDV contributed to data curation; RDV, AP and MDM contributed to writing and original draft preparation; RDV, AP and MDM contributed to writing, review and editing.
| References | ▴Top |
- Roden DM. Mechanisms and management of proarrhythmia. Am J Cardiol. 1998;82(4A):49I-57I.
doi - Goldschlager N, Epstein AE, Naccarelli GV, Olshansky B, Singh B, Collard HR, Murphy E, et al. A practical guide for clinicians who treat patients with amiodarone: 2007. Heart Rhythm. 2007;4(9):1250-1259.
doi pubmed - Wolkove N, Baltzan M. Amiodarone pulmonary toxicity. Can Respir J. 2009;16(2):43-48.
doi pubmed - Lafuente-Lafuente C, Longas-Tejero MA, Bergmann JF, Belmin J. Antiarrhythmics for maintaining sinus rhythm after cardioversion of atrial fibrillation. Cochrane Database Syst Rev. 2012;5:CD005049.
doi - Kober L, Torp-Pedersen C, McMurray JJ, Gotzsche O, Levy S, Crijns H, Amlie J, et al. Increased mortality after dronedarone therapy for severe heart failure. N Engl J Med. 2008;358(25):2678-2687.
doi pubmed - Cardiac Arrhythmia Suppression Trial Investigators. Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. N Engl J Med. 1989;321(6):406-412.
doi pubmed - Pathak RK, Middeldorp ME, Lau DH, Mehta AB, Mahajan R, Twomey D, Alasady M, et al. Aggressive risk factor reduction study for atrial fibrillation and implications for the outcome of ablation: the ARREST-AF cohort study. J Am Coll Cardiol. 2014;64(21):2222-2231.
doi pubmed - Abed HS, Wittert GA, Leong DP, Shirazi MG, Bahrami B, Middeldorp ME, Lorimer MF, et al. Effect of weight reduction and cardiometabolic risk factor management on symptom burden and severity in patients with atrial fibrillation: a randomized clinical trial. JAMA. 2013;310(19):2050-2060.
doi pubmed - Schneider MP, Hua TA, Bohm M, Wachtell K, Kjeldsen SE, Schmieder RE. Prevention of atrial fibrillation by Renin-Angiotensin system inhibition a meta-analysis. J Am Coll Cardiol. 2010;55(21):2299-2307.
doi pubmed - Kirchhof P, Benussi S, Kotecha D, Ahlsson A, Atar D, Casadei B, Castella M, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J. 2016;37(38):2893-2962.
doi pubmed - Duytschaever M, Demolder A, Phlips T, Sarkozy A, El Haddad M, Taghji P, Knecht S, et al. Pulmonary vein isolation with vs. without continued antiarrhythmic drug treatment in subjects with Recurrent Atrial Fibrillation (POWDER AF): results from a multicentre randomized trial. Eur Heart J. 2018;39(16):1429-1437.
doi pubmed - Darkner S, Chen X, Hansen J, Pehrson S, Johannessen A, Nielsen JB, Svendsen JH. Recurrence of arrhythmia following short-term oral amiodarone after catheter ablation for atrial fibrillation: a double-blind, randomized, placebo-controlled study (AMIO-CAT trial). Eur Heart J. 2014;35(47):3356-3364.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


