| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website http://www.cardiologyres.org |
Original Article
Volume 10, Number 3, June 2019, pages 181-187
Clinical Significance of Skin Autofluorescence in Elderly Patients With Long-Standing Persistent Atrial Fibrillation
Takashi Hitsumoto
Hitsumoto Medical Clinic, 2-7-7, Takezakicyou, Shimonoseki City, Yamaguchi 750-0025, Japan
Manuscript submitted May 20, 2019, accepted May 27, 2019
Short title: Skin Autofluorescence and Atrial Fibrillation
doi: https://doi.org/10.14740/cr885
| Abstract | ▴Top |
Background: Recent clinical studies have demonstrated the importance of skin autofluorescence as a cardiovascular risk factor. However, data regarding the relationship between skin autofluorescence and atrial fibrillation are limited. The aim of this study was to clarify the clinical significance of skin autofluorescence in elderly patients with long-standing persistent atrial fibrillation.
Methods: This cross-sectional study enrolled 112 elderly patients with long-standing persistent atrial fibrillation who were treated medically (46 men and 66 women; mean age, 81 ± 9 years). The association between skin autofluorescence and various clinical parameters was examined.
Results: Significant relationships were observed between skin autofluorescence and CHADS2 score (r = 0.53, P < 0.001), high-sensitivity cardiac troponin T level (r = 0.43, P < 0.001), reactive oxygen metabolite levels (r = 0.52, P < 0.001), and whole blood passage time (r = 0.45, P < 0.001). Furthermore, multiple regression analyses showed that these clinical parameters were independent variables when skin autofluorescence was used as a subordinate factor. Receiver-operating characteristic curve analysis indicated that the risk values of skin autofluorescence for high CHADS2 scores (≥ 2) or elevated high-sensitivity cardiac troponin T levels (> 0.014ng/mL) were 2.6 arbitrary units (AU) and 2.7 AU, respectively.
Conclusions: The findings of this study indicated that skin autofluorescence may be a prognostic factor in elderly patients with long-standing persistent atrial fibrillation. The risk value of skin autofluorescence was considered as 2.6 AU or 2.7 AU.
Keywords: Skin autofluorescence; Long-standing persistent atrial fibrillation; CHADS2 score; Blood rheology; High-sensitivity cardiac troponin T; Oxidative stress; Elderly
| Introduction | ▴Top |
Atrial fibrillation is a common cardiovascular disease and is associated with other diseases such as ischemic stroke and heart failure [1]. In recent years, the prevalence of atrial fibrillation has increased because of a concomitant increase in life expectancy [2]. Treatment via an electrophysiologic catheter ablation procedure can be performed for selected patients with atrial fibrillation [3], which may achieve a complete cure in some. Catheter ablation is particularly useful in patients with paroxysmal atrial fibrillation, but less so in those with persistent atrial fibrillation [4, 5]. In addition, elderly patients with persistent atrial fibrillation are less likely to receive the procedure for various reasons such as duration of atrial fibrillation, symptoms, activities of daily practice, patient refusal, and limited procedural benefit.
Advanced glycation end products (AGEs) and receptors of AGEs play an important role in the pathophysiology of cardiovascular disease [6, 7]. Among the methods used to evaluate AGEs, skin autofluorescence is known to be a simple and reliable marker of AGEs in vivo, and recent clinical studies have indicated that skin autofluorescence is significantly associated with cardiovascular disease [8-10]. However, data regarding the relationship between skin autofluorescence and atrial fibrillation are limited. This cross-sectional study attempted to clarify the clinical significance of skin autofluorescence in elderly patients with long-standing persistent atrial fibrillation.
| Materials and Methods | ▴Top |
Patients
In this study, 112 elderly outpatients (age ≥ 65 years) with long-standing persistent atrial fibrillation who were being treated medically at the Hitsumoto Medical Clinic, Yamaguchi, Japan were enrolled between January 2017 and December 2018. Long-standing persistent atrial fibrillation (presence of atrial fibrillation for ≥ 1 year after initiation of rhythm control treatment) is defined by the 2010 Guidelines for the Management of Atrial Fibrillation of the European Society of Cardiology [11]. The patients included 46 (41%) men and 66 (59%) women. The mean patient age was 81 ± 9 years. The study was approved by the Institutional Review Board of the Hitsumoto Medical Clinic (approval number 2017-01) and was conducted in compliance with the Declaration of Helsinki. All patients provided informed consent.
Measurement of skin autofluorescence
Skin autofluorescence was measured using a commercial device (AGE Reader™; DiagnOptics, Groningen, the Netherlands), as previously described [12, 13]. Autofluorescence was defined as the average light intensity per nanometer between 300 and 420 nm. Skin autofluorescence levels were expressed in arbitrary units (AU). All measurements were performed on the volar side of the forearm approximately 10 - 15 cm below the elbow while the patients were in the sitting position. The value of pentosidine, a major component of AGEs, has been previously measured using skin biopsy on the volar side of the forearm and seemed to correlate with skin autofluorescence [14]. Previous reports have established the validity and reliability of skin autofluorescence level measurements using this method in the Japanese population [13].
Evaluation of clinical parameters
Various clinical parameters were evaluated, including classic risk factors of cardiovascular disease, such as CHADS2 score [15], echocardiographic findings, kidney function, brain natriuretic peptide level, high-sensitivity cardiac troponin T (hs-cTnT) level, reactive oxygen metabolites (a marker of oxidative stress), and whole blood passage time (a marker of blood rheology). Obesity was identified using body mass index, calculated as the weight (kg) divided by the squared height (m2). Current smoking was defined as smoking at least one cigarette per day over the previous 28 days. Hypertension was defined as systolic blood pressure ≥ 140 mm Hg, diastolic blood pressure ≥ 90 mm Hg, or the use of antihypertensive medication. Dyslipidemia was defined as low-density lipoprotein cholesterol level ≥ 140 mg/dL, high-density lipoprotein cholesterol level ≤ 40 mg/dL, triglyceride level ≥ 150 mg/dL, or the use of lipid-lowering medication. Diabetes mellitus was defined as fasting blood glucose level ≥ 126 mg/dL, hemoglobin A1c (HbA1c) level ≥ 6.5%, or the use of antidiabetic medication or exogenous insulin. Standard echocardiography was performed using the HI VISION Avius ultrasound system (Hitachi Medical Corporation, Tokyo, Japan). Left ventricular wall thickness, left ventricular end-diastolic diameter, left ventricular ejection fraction, and left atrial dimension were measured by echocardiography. The estimated glomerular filtration rate was calculated using the adjusted Modification of Diet in Renal Disease Study equation, which was proposed by the working group of the Japanese Chronic Kidney Disease Initiative [16]. Brain natriuretic peptide levels were measured using the SHIONOSPOT® Reader (Shionogi & Co., Osaka, Japan), and hs-cTnT levels were measured using an hs-cTnT assay (Roche Diagnostics, Basel, Switzerland) [17]. As an in vivo oxidative stress marker [18], the reactive oxygen metabolites (d-ROMs) test was performed (Diacron, Grosseto, Italy). Blood rheology was evaluated by measuring whole blood passage time with an MC-FAN HR300 rheometer (MC Healthcare, Tokyo, Japan), as previously reported [19, 20].
Statistical analysis
In this study, data were expressed as mean ± standard deviation. Comparisons were made using the Student’s t-test. Simple regression analysis was performed using the Pearson or Spearman rank correlation, and a multivariate analysis was performed using multiple regression. Receiver-operating characteristic curve analysis was performed, and the maximum Youden’s index [21] was used to determine the optimal skin autofluorescence cutoff values for high CHADS2 scores and high hs-cTnT levels. P < 0.05 was considered to be statistically significant. StatView J5.0 (HULINKS, Tokyo, Japan) and MedCalc for Windows (MedCalc Software, Ostend, Belgium) software were used for analysis.
| Results | ▴Top |
Table 1 summarizes the patients’ characteristics. The overall mean skin autofluorescence was 2.8 ± 0.5 AU (range, 2.0 - 4.6 AU). Eighty-six patients (77%) had a CHADS2 score ≥ 2. Figure 1 shows the significant positive correlation between skin autofluorescence and the CHADS2 score. Figure 2 shows the relationship between skin autofluorescence and the components of the CHADS2 score. Skin autofluorescence had a significant correlation with the following components: congestive heart failure, age, diabetes mellitus, and stroke/transient ischemic attack. Table 2 presents the correlation between skin autofluorescence and various clinical parameters. Smoking status, HbA1c, estimated glomerular filtration rate, brain natriuretic peptide level, hs-cTnT level, d-ROMs test, whole blood passage time, and renin-angiotensin inhibitor use significantly correlated with skin autofluorescence. Table 3 summarizes the results of multiple regression analysis with skin autofluorescence as a subordinate factor. Explanatory factors were selected by examining the multicollinearity among the variables using stepwise regression; four factors (CHADS2 score, whole blood passage time, d-ROMs test, and hs-cTnT level) were identified as independent variables. Figure 3 shows the receiver-operating characteristic curve analysis for the detection of high CHADS2 scores (≥ 2) and elevated hs-cTnT levels (> 0.014 ng/mL), based on previous studies [22, 23]. The maximum Youden’s index indicated that the optimal cutoff values of skin autofluorescence to determine high CHADS2 scores and elevated hs-cTnT levels were 2.6 AU and 2.7 AU, respectively.
 Click to view | Table 1. Patient Characteristics |
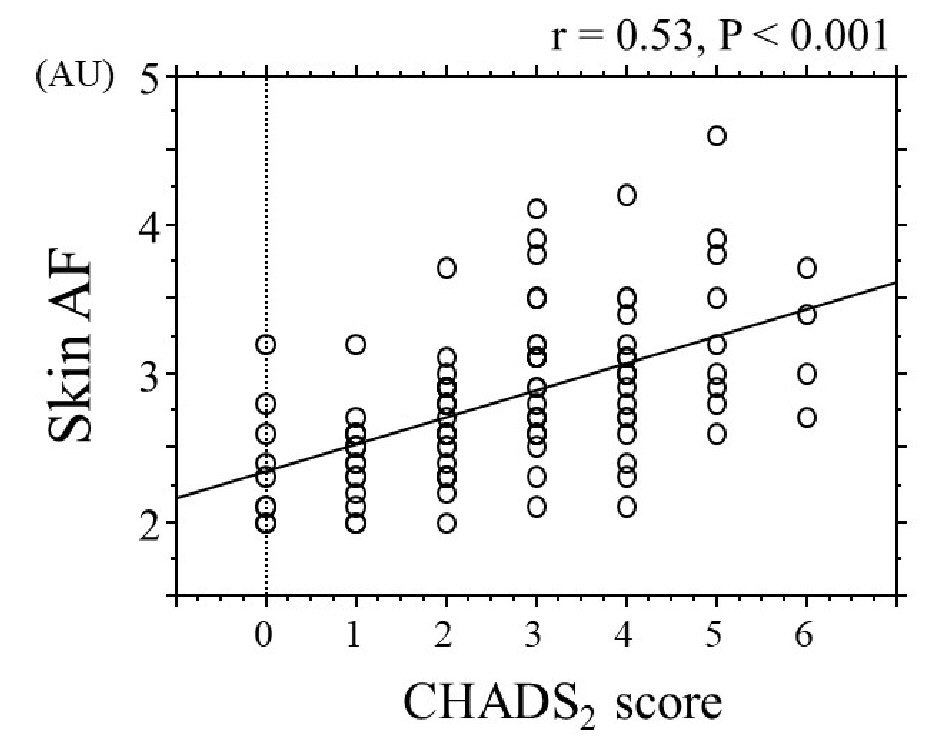 Click for large image | Figure 1. Correlation between skin autofluorescence and CHADS2 score. AF: autofluorescence; AU: arbitrary unit. |
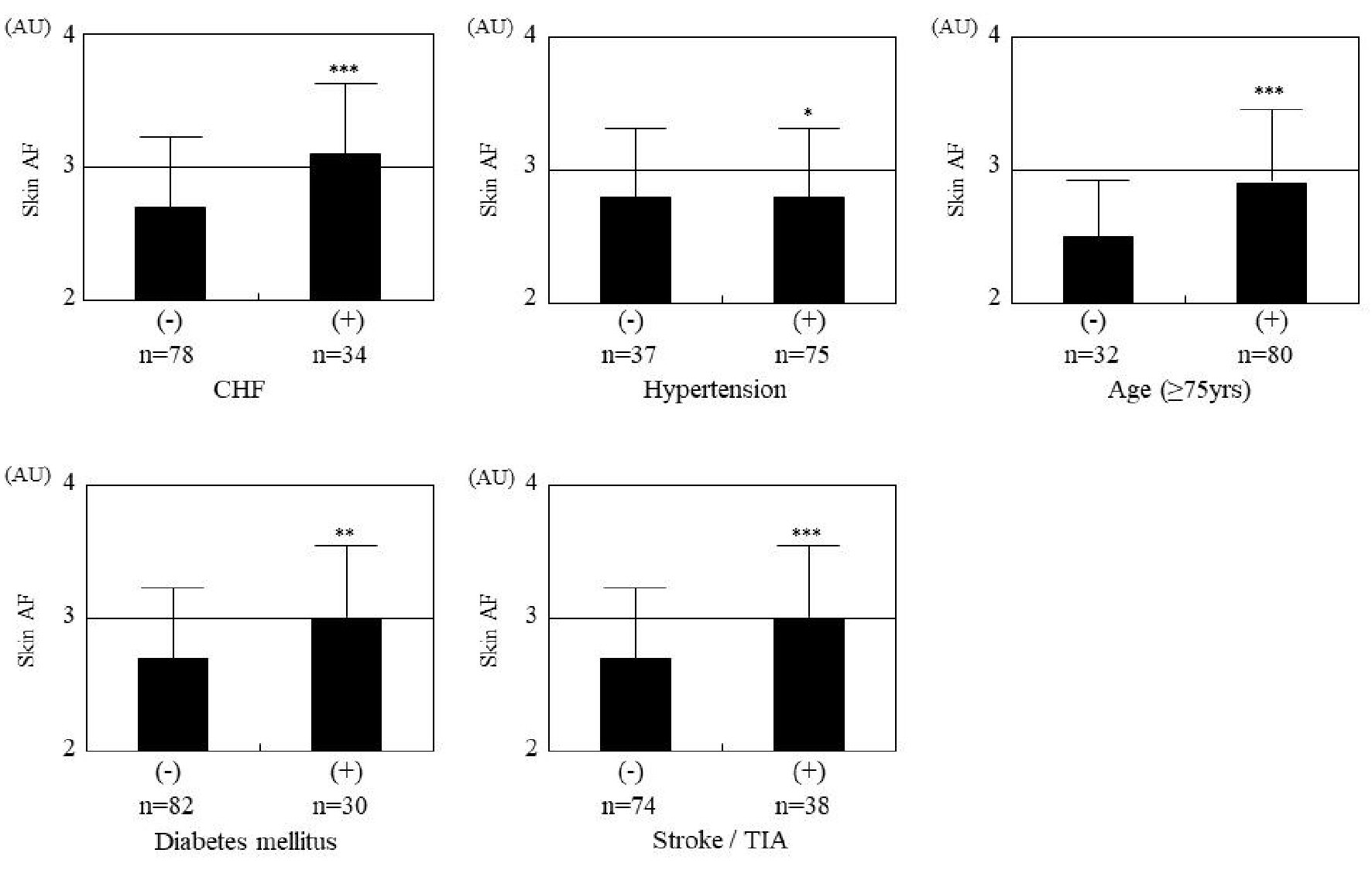 Click for large image | Figure 2. Relationship between skin autofluorescence and components of the CHADS2 score. Comparisons are between those with and without the following factors: CHF (3.1 ± 0.5 AU vs. 2.7 ± 0.5 AU, respectively, P < 0.001), age ≥ 75 years (2.9 ± 0.5 AU vs. 2.5 ± 0.4 AU, respectively, P < 0.001), diabetes mellitus (3.0 ± 0.5 AU vs. 2.7 ± 0.5 AU, respectively, P = 0.019), and stroke/TIA (3.0 ± 0.5 AU vs. 2.7 ± 0.5 AU, respectively, P < 0.001). *P = 0.598, **P = 0.019, ***P < 0.001. AF: autofluorescence; AU: arbitrary units; CHF: congestive heart failure; TIA: transient ischemic attack. |
 Click to view | Table 2. Relationship Between Skin Autofluorescence and Various Clinical Parameters |
 Click to view | Table 3. Multiple Regression Analysis for Skin Autofluorescence |
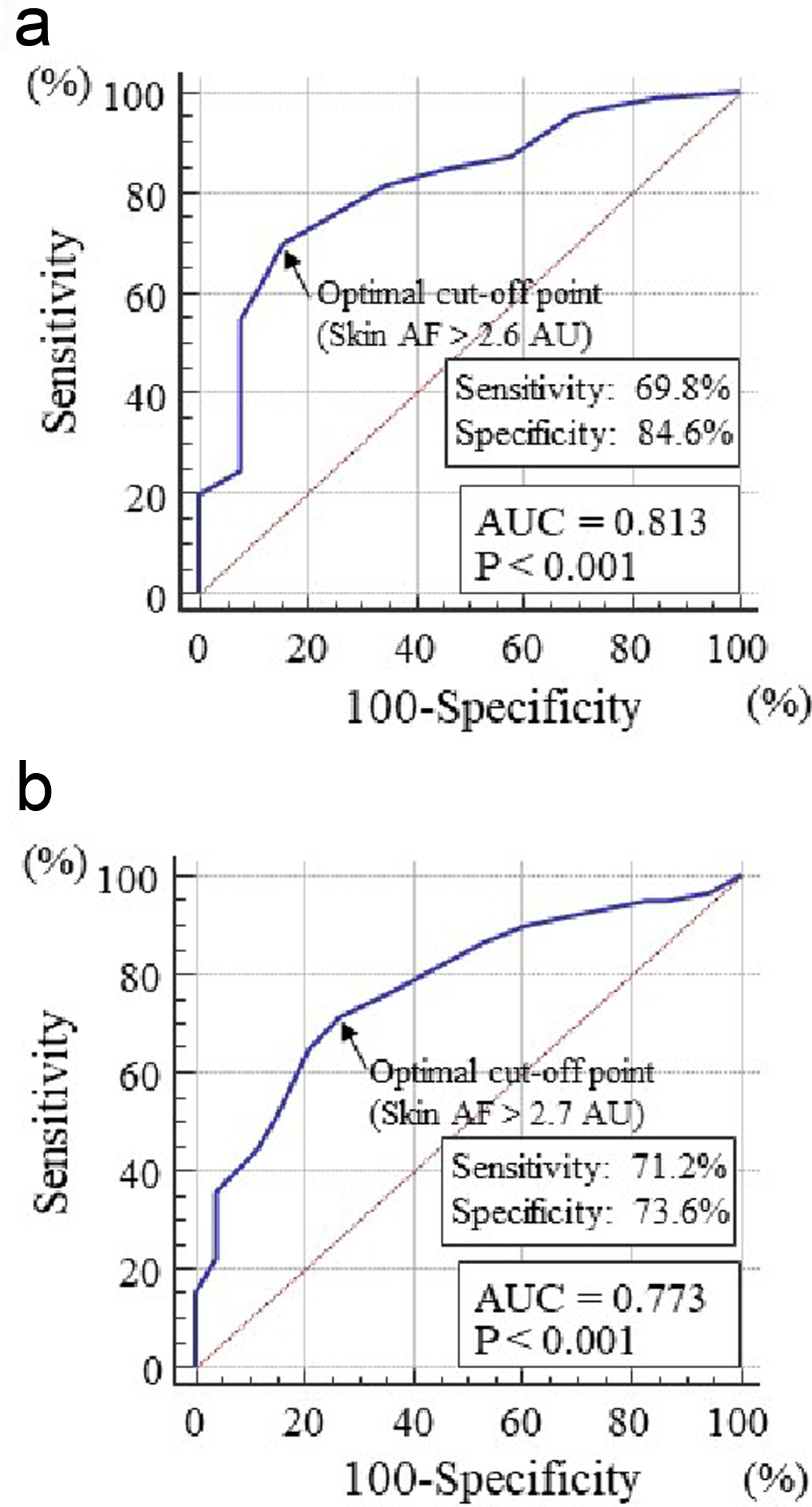 Click for large image | Figure 3. Receiver-operating characteristic curve analysis for the detection of high CHADS2 scores or elevated hs-cTnT levels based on skin autofluorescence. The maximum Youden’s index indicated that skin autofluorescence values > 2.6 AU and > 2.7 AU are the optimal cutoff points to identify high CHADS2 scores (≥ 2) or elevated hs-cTnT levels (> 0.014 ng/mL), respectively. (a) CHADS2 score. (b) hs-cTnT levels. AF: autofluorescence; AU: arbitrary unit; hs-cTnT: high-sensitivity cardiac troponin T; AUC: area under the curve. |
| Discussion | ▴Top |
This study aimed to clarify the clinical significance of skin autofluorescence in elderly patients with long-standing persistent atrial fibrillation. The results showed an independent association between skin autofluorescence and CHADS2 score, whole blood passage time, hs-cTnT level, and d-ROMs test. In addition, the receiver-operating characteristic curve analysis indicated that the skin autofluorescence values associated with high CHADS2 scores and elevated hs-cTnT levels in this study population were > 2.6 AU and > 2.7 AU, respectively.
The CHADS2 score is a well-known predictor of ischemic stroke in patients with atrial fibrillation [15, 22]. Therefore, the independent association between skin autofluorescence and the CHADS2 score in this study suggests that increased skin autofluorescence is closely associated with ischemic stroke incidence. Although several physiologic processes are involved in ischemic stroke, vascular dysfunction is one of the main causes. Basic science studies have reported that AGEs or receptors of AGEs can induce vascular cell dysfunction [24-26]. In addition, clinical studies have indicated a significant association between skin autofluorescence and pulsatility index at the common carotid artery, which reflects vascular resistance in cerebral vessels [27]. Thus, the relation of ischemic stroke and skin autofluorescence can be partly explained by vascular dysfunction in cerebral vessels caused by AGEs. Another mechanism of ischemic stroke is cerebral embolism, which is closely associated with atrial fibrillation. In this clinical setting, thrombosis is known to be produced mainly in the left atrium or left atrial appendage, where impaired blood rheology has been shown to be an important factor [28, 29]. Basic science investigations have indicated that AGEs cause impaired blood rheology by mechanisms such as leukocyte-endothelial interaction, activation of platelet aggregation, and increased levels of plasminogen activator inhibitor-1 [30-32]. Therefore, the independent association between skin autofluorescence and whole blood passage time in this study can be explained by hypothesizing that AGEs play an important role in impairing blood rheology in elderly patients with long-standing persistent atrial fibrillation; consequently, treatment directed at decreasing AGEs may prevent ischemic stroke by improving blood rheology.
Recent clinical studies have shown the clinical importance of hs-cTnT levels as a prognostic factor in patients with atrial fibrillation [23, 33]. In addition, hs-cTnT is used as a biomarker to clinically evaluate the severity of myocardial injury. Hofmann et al found a significant relationship between AGE-modified cardiac tissue collagen and skin autofluorescence [34]. They also showed that the AGE level found at the volar side of the forearm seemed to reflect the degree of AGE accumulation in cardiomyocytes. In addition, basic science studies have indicated that AGEs or receptors of AGEs could influence myocardial injury via several pathways [35, 36]. Thus, the results of this study and previous ones suggest that AGEs play a crucial role in the progression of myocardial damage in elderly patients with long-standing persistent atrial fibrillation.
Several researchers have emphasized the importance of oxidative stress in the pathogenesis of atrial fibrillation [37, 38], and a number of basic science and clinical studies have shown a close association between AGEs or receptors of AGEs and oxidative stress in the heart and arterial vessels [24, 39, 40]. The results of this study also indicated that the d-ROMs test as an in vivo marker of oxidative stress is an important factor for skin autofluorescence. A previous study reported that increased activity of the renin-angiotensin system caused increased oxidative stress or AGE production, and the use of an angiotensin receptor blocker decreased both oxidative stress and receptors of AGEs [41]. This study showed a significantly negative association between angiotensin receptor blocker use and skin autofluorescence, even though angiotensin receptor blocker use was not selected in the multivariate model. Therefore, we have started to intervene by prescribing an angiotensin receptor blocker for patients with high skin autofluorescence; consequently, we expect a reduction in cardiovascular events, including ischemic stroke or heart failure, in elderly patients with long-standing persistent atrial fibrillation.
This study clarified the clinical usefulness of assessing skin autofluorescence to detect a high CHADS2 score ≥ 2 or an elevated hs-cTnT level > 0.014 ng/mL, which are associated with cardiovascular events such as ischemic stroke, heart failure, and coronary artery disease in patients with atrial fibrillation according to previous reports. The receiver-operating characteristic curve analysis indicated that skin autofluorescence values > 2.6 AU and > 2.7 AU are the optimal cutoff points to identify a high CHADS2 score and an elevated hs-cTnT level, respectively. Therefore, this study indicated that maintaining skin autofluorescence values ≤ 2.6 AU or ≤ 2.7 AU in elderly patients with long-standing persistent atrial fibrillation may decrease cardiovascular events. Genevieve et al performed a study regarding the association between skin autofluorescence and HbA1c levels in patients with diabetes mellitus, and reported that skin autofluorescence was significantly associated with the means of the last five and 10 HbA1c values [42]. In addition, Isami et al reported that lifestyle habits such as physical activity, nonsmoking, adequate sleep, low mental stress level, eating breakfast, and abstaining from sugary foods were independently associated with lower skin autofluorescence [43]. Therefore, it appears that long-term adequate blood glucose control and good lifestyle habits are important to maintain lower skin autofluorescence as early as possible.
Limitations
This study has several limitations. First, the various medical treatments may have affected the study results. Second, skin autofluorescence was measured in only Japanese patients; previous studies have indicated that skin autofluorescence varies according to race [44, 45]. Therefore, the cutoff values for skin autofluorescence found in this study may not apply to non-Japanese populations. Finally, the study design was a single-center cross-sectional study, and the sample size was relatively small. Additional prospective studies, including evaluations of interventional therapies, are required to clarify the clinical significance of skin autofluorescence in elderly patients with long-standing persistent atrial fibrillation.
Conclusions
In conclusion, the findings of this study showed that skin autofluorescence may be a prognostic factor in elderly patients with long-standing persistent atrial fibrillation. The risk value of skin autofluorescence was considered as 2.6 AU or 2.7 AU. Further prospective studies that include the evaluation of therapies are required to validate the results of this study.
Acknowledgments
The author is grateful to the individuals who participated in this study.
Financial Disclosure
None to declare.
Conflict of Interest
None to declare.
Informed Consent
All patients provided informed consent.
Author Contributions
The author was involved in preparing the study design as well as in the acquisition, analysis, and interpretation of data.
| References | ▴Top |
- Stewart S, Hart CL, Hole DJ, McMurray JJ. A population-based study of the long-term risks associated with atrial fibrillation: 20-year follow-up of the Renfrew/Paisley study. Am J Med. 2002;113(5):359-364.
doi - Akao M, Chun YH, Wada H, Esato M, Hashimoto T, Abe M, Hasegawa K, et al. Current status of clinical background of patients with atrial fibrillation in a community-based survey: the Fushimi AF Registry. J Cardiol. 2013;61(4):260-266.
doi pubmed - Haissaguerre M, Jais P, Shah DC, Garrigue S, Takahashi A, Lavergne T, Hocini M, et al. Electrophysiological end point for catheter ablation of atrial fibrillation initiated from multiple pulmonary venous foci. Circulation. 2000;101(12):1409-1417.
doi pubmed - Tilz RR, Rillig A, Thum AM, Arya A, Wohlmuth P, Metzner A, Mathew S, et al. Catheter ablation of long-standing persistent atrial fibrillation: 5-year outcomes of the Hamburg Sequential Ablation Strategy. J Am Coll Cardiol. 2012;60(19):1921-1929.
doi pubmed - Gaita F, Caponi D, Scaglione M, Montefusco A, Corleto A, Di Monte F, Coin D, et al. Long-term clinical results of 2 different ablation strategies in patients with paroxysmal and persistent atrial fibrillation. Circ Arrhythm Electrophysiol. 2008;1(4):269-275.
doi pubmed - Yamagishi S, Nakamura N, Suematsu M, Kaseda K, Matsui T. Advanced glycation end products: a molecular target for vascular complications in diabetes. Mol Med. 2015;21(Suppl 1):S32-40.
doi pubmed - Koike S, Yano S, Tanaka S, Sheikh AM, Nagai A, Sugimoto T. Advanced glycation end-products induce apoptosis of vascular smooth muscle cells: a mechanism for vascular calcification. Int J Mol Sci. 2016;17(9):1567.
doi pubmed - Schmidt AM. Skin autofluorescence, 5-year mortality, and cardiovascular events in peripheral arterial disease: all that glitters is surely not gold. Arterioscler Thromb Vasc Biol. 2014;34(4):697-699.
doi pubmed - Furuya F, Shimura H, Takahashi K, Akiyama D, Motosugi A, Ikegishi Y, Haraguchi K, et al. Skin autofluorescence is a predictor of cardiovascular disease in chronic kidney disease patients. Ther Apher Dial. 2015;19(1):40-44.
doi pubmed - Hitsumoto T. Clinical Significance of Skin Autofluorescence in Patients With Type 2 Diabetes Mellitus With Chronic Heart Failure. Cardiol Res. 2018;9(2):83-89.
doi pubmed - Camm AJ, Kirchhof P, Lip GY, Schotten U, Savelieva I, Ernst S, Van Gelder IC, et al. Guidelines for the management of atrial fibrillation: the Task Force for the Management of Atrial Fibrillation of the European Society of Cardiology (ESC). Europace. 2010;12(10):1360-1420.
doi pubmed - Meerwaldt R, Hartog JW, Graaff R, Huisman RJ, Links TP, den Hollander NC, Thorpe SR, et al. Increased accumulation of skin advanced glycation end-products precedes and correlates with clinical manifestation of diabetic neuropathy. J Am Soc Nephrol. 2005;16(12):3687-3693.
doi pubmed - Masterson TD, Gilbert-Diamond D, Lansigan RK, Kim SJ, Schiffelbein JE, Emond JA. Measurement of external food cue responsiveness in preschool-age children: Preliminary evidence for the use of the external food cue responsiveness scale. Appetite. 2019;139:119-126.
doi pubmed - Meerwaldt R, Graaff R, Oomen PHN, Links TP, Jager JJ, Alderson NL, Thorpe SR, et al. Simple non-invasive assessment of advanced glycation endproduct accumulation. Diabetologia. 2004;47(7):1324-1330.
doi pubmed - Gage BF, Waterman AD, Shannon W, Boechler M, Rich MW, Radford MJ. Validation of clinical classification schemes for predicting stroke: results from the National Registry of Atrial Fibrillation. JAMA. 2001;285(22):2864-2870.
doi pubmed - Imai E, Horio M, Nitta K, Yamagata K, Iseki K, Hara S, Ura N, et al. Estimation of glomerular filtration rate by the MDRD study equation modified for Japanese patients with chronic kidney disease. Clin Exp Nephrol. 2007;11(1):41-50.
doi pubmed - Mingels A, Jacobs L, Michielsen E, Swaanenburg J, Wodzig W, van Dieijen-Visser M. Reference population and marathon runner sera assessed by highly sensitive cardiac troponin T and commercial cardiac troponin T and I assays. Clin Chem. 2009;55(1):101-108.
doi pubmed - Cesarone MR, Belcaro G, Carratelli M, Cornelli U, De Sanctis MT, Incandela L, Barsotti A, et al. A simple test to monitor oxidative stress. Int Angiol. 1999;18(2):127-130.
- Kikuchi Y, Sato K, Mizuguchi Y. Modified cell-flow microchannels in a single-crystal silicon substrate and flow behavior of blood cells. Microvasc Res. 1994;47(1):126-139.
doi pubmed - Hitsumoto T. Clinical impact of hemorheology on subclinical myocardial injury in patients with hypertension. J Clin Med Res. 2018;10(12):928-935.
doi pubmed - Schisterman EF, Perkins NJ, Liu A, Bondell H. Optimal cut-point and its corresponding Youden Index to discriminate individuals using pooled blood samples. Epidemiology. 2005;16(1):73-81.
doi pubmed - Suzuki S, Yamashita T, Okumura K, Atarashi H, Akao M, Ogawa H, Inoue H. Incidence of ischemic stroke in Japanese patients with atrial fibrillation not receiving anticoagulation therapy—pooled analysis of the Shinken Database, J-RHYTHM Registry, and Fushimi AF Registry. Circ J. 2015;79(2):432-438.
doi pubmed - Stoyanov KM, Giannitsis E, Biener M, Mueller-Hennessen M, Arens K, Katus HA, Vafaie M. Prognostic value of elevated high-sensitivity cardiac troponin T in patients admitted to an emergency department with atrial fibrillation. Europace. 2018;20(4):582-588.
doi pubmed - Yamagishi S, Imaizumi T. Diabetic vascular complications: pathophysiology, biochemical basis and potential therapeutic strategy. Curr Pharm Des. 2005;11(18):2279-2299.
doi pubmed - Chang JS, Wendt T, Qu W, Kong L, Zou YS, Schmidt AM, Yan SF. Oxygen deprivation triggers upregulation of early growth response-1 by the receptor for advanced glycation end products. Circ Res. 2008;102(8):905-913.
doi pubmed - Brett J, Schmidt AM, Yan SD, Zou YS, Weidman E, Pinsky D, Nowygrod R, et al. Survey of the distribution of a newly characterized receptor for advanced glycation end products in tissues. Am J Pathol. 1993;143(6):1699-1712.
- Hitsumoto T. Impact of Hemorheology Assessed by the Microchannel Method on Pulsatility Index of the Common Carotid Artery in Patients With Type 2 Diabetes Mellitus. J Clin Med Res. 2017;9(7):579-585.
doi pubmed - Siostrzonek P, Koppensteiner R, Gossinger H, Zangeneh M, Heinz G, Kreiner G, Stumpflen A, et al. Hemodynamic and hemorheologic determinants of left atrial spontaneous echo contrast and thrombus formation in patients with idiopathic dilated cardiomyopathy. Am Heart J. 1993;125(2 Pt 1):430-434.
doi - Leithauser B, Jung F, Park JW. Rheological and hemostasiological aspects of thrombus formation in the left atrial appendage in atrial fibrillation? A new strategy for prevention of cardioembolic stroke. Clin Hemorheol Microcirc. 2010;45(2-4):311-323.
- Morigi M, Angioletti S, Imberti B, Donadelli R, Micheletti G, Figliuzzi M, Remuzzi A, et al. Leukocyte-endothelial interaction is augmented by high glucose concentrations and hyperglycemia in a NF-kB-dependent fashion. J Clin Invest. 1998;101(9):1905-1915.
doi pubmed - Hasegawa Y, Suehiro A, Higasa S, Namba M, Kakishita E. Enhancing effect of advanced glycation end products on serotonin-induced platelet aggregation in patients with diabetes mellitus. Thromb Res. 2002;107(6):319-323.
doi - Fishman SL, Sonmez H, Basman C, Singh V, Poretsky L. The role of advanced glycation end-products in the development of coronary artery disease in patients with and without diabetes mellitus: a review. Mol Med. 2018;24(1):59.
doi pubmed - Hijazi Z, Siegbahn A, Andersson U, Lindahl B, Granger CB, Alexander JH, Atar D, et al. Comparison of cardiac troponins I and T measured with high-sensitivity methods for evaluation of prognosis in atrial fibrillation: an ARISTOTLE substudy. Clin Chem. 2015;61(2):368-378.
doi pubmed - Hofmann B, Jacobs K, Navarrete Santos A, Wienke A, Silber RE, Simm A. Relationship between cardiac tissue glycation and skin autofluorescence in patients with coronary artery disease. Diabetes Metab. 2015;41(5):410-415.
doi pubmed - Ma H, Li SY, Xu P, Babcock SA, Dolence EK, Brownlee M, Li J, et al. Advanced glycation endproduct (AGE) accumulation and AGE receptor (RAGE) up-regulation contribute to the onset of diabetic cardiomyopathy. J Cell Mol Med. 2009;13(8B):1751-1764.
doi pubmed - Bucciarelli LG, Ananthakrishnan R, Hwang YC, Kaneko M, Song F, Sell DR, Strauch C, et al. RAGE and modulation of ischemic injury in the diabetic myocardium. Diabetes. 2008;57(7):1941-1951.
doi pubmed - Samman Tahhan A, Sandesara PB, Hayek SS, Alkhoder A, Chivukula K, Hammadah M, Mohamed-Kelli H, et al. Association between oxidative stress and atrial fibrillation. Heart Rhythm. 2017;14(12):1849-1855.
doi pubmed - Korantzopoulos P, Letsas K, Fragakis N, Tse G, Liu T. Oxidative stress and atrial fibrillation: an update. Free Radic Res. 2018;52(11-12):1199-1209.
doi pubmed - Bodiga VL, Eda SR, Bodiga S. Advanced glycation end products: role in pathology of diabetic cardiomyopathy. Heart Fail Rev. 2014;19(1):49-63.
doi pubmed - Yu Y, Wang L, Delguste F, Durand A, Guilbaud A, Rousselin C, Schmidt AM, et al. Advanced glycation end products receptor RAGE controls myocardial dysfunction and oxidative stress in high-fat fed mice by sustaining mitochondrial dynamics and autophagy-lysosome pathway. Free Radic Biol Med. 2017;112:397-410.
doi pubmed - Yamagishi S, Nakamura K, Matsui T. Potential utility of telmisartan, an angiotensin II type 1 receptor blocker with peroxisome proliferator-activated receptor-gamma (PPAR-gamma)-modulating activity for the treatment of cardiometabolic disorders. Curr Mol Med. 2007;7(5):463-469.
doi pubmed - Genevieve M, Vivot A, Gonzalez C, Raffaitin C, Barberger-Gateau P, Gin H, Rigalleau V. Skin autofluorescence is associated with past glycaemic control and complications in type 1 diabetes mellitus. Diabetes Metab. 2013;39(4):349-354.
doi pubmed - Isami F, West BJ, Nakajima S, Yamagishi SI. Association of advanced glycation end products, evaluated by skin autofluorescence, with lifestyle habits in a general Japanese population. J Int Med Res. 2018;46(3):1043-1051.
doi pubmed - Simon Klenovics K, Kollarova R, Hodosy J, Celec P, Sebekova K. Reference values of skin autofluorescence as an estimation of tissue accumulation of advanced glycation end products in a general Slovak population. Diabet Med. 2014;31(5):581-585.
doi pubmed - Ahmad MS, Kimhofer T, Ahmad S, AlAma MN, Mosli HH, Hindawi SI, Mook-Kanamori DO, et al. Ethnicity and skin autofluorescence-based risk-engines for cardiovascular disease and diabetes mellitus. PLoS One. 2017;12(9):e0185175.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


