| Cardiology Research, ISSN 1923-2829 print, 1923-2837 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, Cardiol Res and Elmer Press Inc |
| Journal website http://www.cardiologyres.org |
Case Report
Volume 11, Number 2, April 2020, pages 118-128
Erdheim-Chester Disease With Extensive Pericardial Involvement: A Case Report and Systematic Review
Alejandro Sanchez-Nadalesa, c, Andrea Anampa-Guzmanb, Jessica Navarro-Mottaa
aDepartment of Medicine, Advocate Illinois Masonic Medical Center, Chicago, IL, USA
bSociedad Cientifica de San Fernando, Faculty of Medicine, Universidad Nacional Mayor de San Marcos, Lima, Peru
cCorresponding Author: Alejandro Sanchez-Nadales, Department of Medicine, Advocate Illinois Masonic Medical Center, Chicago, IL, USA
Manuscript submitted February 9, 2020, accepted February 26, 2020
Short title: ECD With Pericardial Involvement
doi: https://doi.org/10.14740/cr1025
| Abstract | ▴Top |
Erdheim-Chester disease (ECD) is a rare non-Langerhans cell histiocytosis characterized by systemic xanthogranulomatous infiltration. We described the case of a female adult presenting with pericardial effusion. Pericardial infiltration is the most frequent cardiac manifestation of ECD and is the one discussed in this article. We found that the majority of patients with pericardial infiltration needed a cardiovascular procedure.
Keywords: ECD; Pericardial infiltration; BRAF mutation; Vemurafenib
| Introduction | ▴Top |
Erdheim-Chester disease (ECD) is a rare multisystem non-Langerhans cell histiocytosis characterized by systemic xanthogranulomatous infiltration from histiocytes [1, 2]. This disease was first described by Erdheim and Chester in 1930 [3]. The number of cases reporting this disease is small (less than 600), but it has had dramatically increased in recent years, maybe by the increased diagnostic methods available, which enhances the recognition of the disease [4]. Patients with ECD commonly present in the fifth decade; however, pediatric and geriatric presentations have also been reported. ECD results from xanthogranulomatous infiltration of foamy histiocytes; these cells are positive for CD68, negative for CD1a, and in 80% of the cases negative for S100 [5]. ECD manifests as long bone infiltrations, exophthalmos, diabetes insipidus, xanthelasma, interstitial lung disease, bilateral adrenal infiltration, retroperitoneal fibrosis with perirenal or ureteral infiltration causing hydronephrosis and renal failure, testicular infiltration, and in exceptional involves the hearth and the central nervous system [6].
The diagnosis of ECD is based on radiologic and histologic criteria. Imaging studies are prompted for the bone pain referred by the patients. X-rays show bilateral symmetric metaphyseal osteosclerosis of long bones. These lesions are considered virtually pathognomonic. The 99mTc Bone Scintigraphy shows symmetric and abnormally strong 99mTc labeling of the distal ends of the long bones. Microscopy of the lesions shows typical ECD histiocytes. These histiocytes are non-Langerhans’ foamy histiocytes, which lack Birbeck granules, nested within a polymorphic granuloma, fibrosis or xanthogranulomatous. Pathological confirmation of CD68+, CD1a- histiocytes is both sufficient and mandatory for the diagnosis of ECD [6].
Specific somatic genetic mutations in the BRAF V600 gene have been reported in around 54% of patients suffering of ECD, rendering a therapeutic target for the kinase inhibitor, vemurafenib [7]. This medication is the only active Food and Drug Administration (FDA) approved treatment for ECD and has demonstrated long-term efficacy in patients with this genetic mutation [8]. Other medications, as dabrafenib (a BRAF V600E inhibitor) and trametinib (a mitogen-activated protein kinase inhibitor), are under study as potential therapeutic options [9].
| Case Report | ▴Top |
We present the case of a 53-year-old woman with diabetes insipidus that has been treated with desmopressin for about 8 years, migraine with aura, and known ECD confirmed by biopsy, with possible involvement of the great cardiac vessel and infiltrative cardiomyopathy. Over the past several years, she has dealt with a multitude of intermittently exacerbated symptoms, including headaches, fatigue, lethargy, exertional dyspnea, flushing, and peripheral edema, which have been clinically attributed to mast cell activation. She was admitted twice during the previous year with symptoms of fluid retention that resolved with diuresis. She was diagnosed with ECD 2 months before this admission, with biopsy confirmation from the vertebra and the left femur. She was admitted to our institution for further management of a newly identified large pericardial effusion (Fig. 1) and ascites, accurately to assess the extension of the cardiac involvement by this disease. In the interim, she had complained of intermittent midsternal chest discomfort associated with dry cough and palpitations that worsened while lying flat.
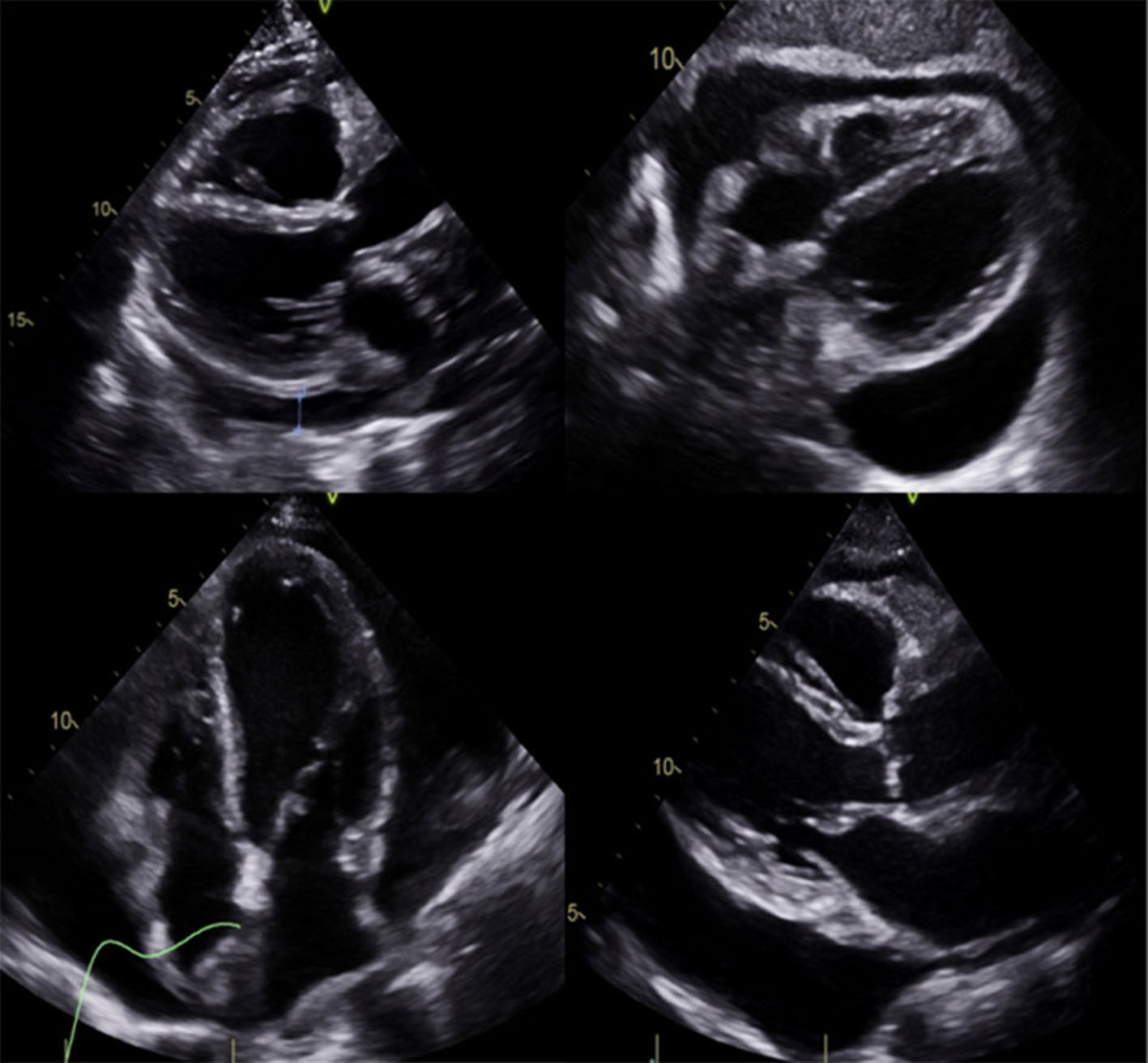 Click for large image | Figure 1. Large pericardial effusion. |
She was found to have an acute right frontal infarct and multifocal stenosis through the vessel imaging modalities. A magnetic resonance imaging (MRI) showed infarction in the right frontal and parietal white matter, while the magnetic resonance angiography (MRA) revealed extensive intracranial stenosis in the anterior circulation. The final cerebral angiogram showed a right internal carotid artery occlusion, likely secondary to fibromuscular dysplasia changes (Fig. 2). Cerebrospinal fluid tests were normal, and the neurology service concluded that these changes were consistent with vasculopathy secondary to ECD with BRAF mutation.
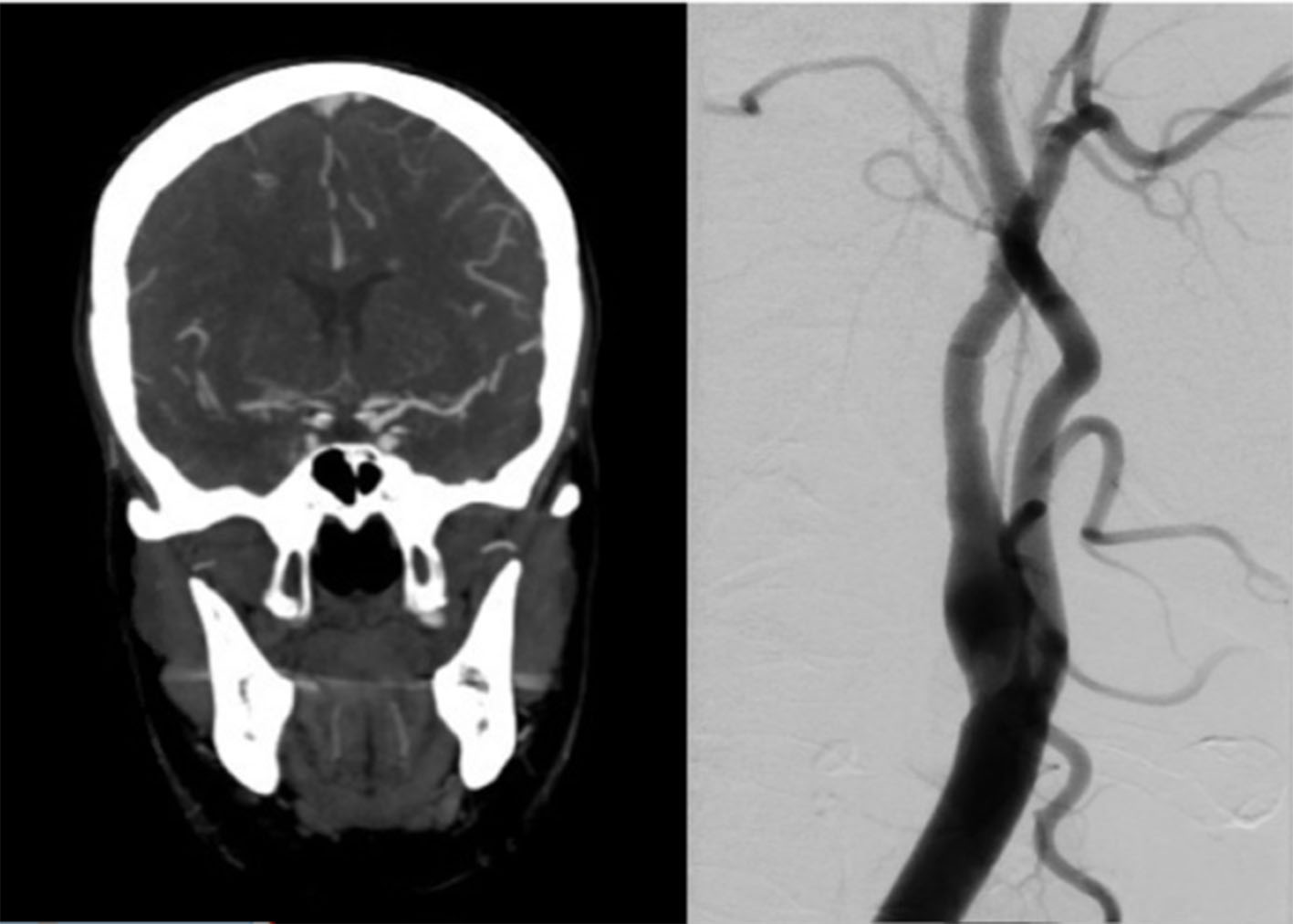 Click for large image | Figure 2. Right internal carotid artery occlusion. |
Transesophageal echocardiography (TEE) showed a large circumferential pericardial effusion greater than 2 cm with maximum thickness around the inferior and posterior left ventricle wall with the partial systolic collapse of the right atrium. She had an ejection fraction of 69% and large pericardial effusion without tamponade physiology. After the evacuation of the pericardial effusion via video-assisted thoracoscopic surgery (VATS), there was minimal pericardial effusion remaining. The pathology exam from pericardium showed histiocytic infiltration compatible with ECD, positive for the BRAF mutation. From these findings, it was concluded that the patient was suffering from a vasculopathy related to ECD. She received corticosteroids for the treatment of the histiocytosis caused by ECD.
She continued her desmopressin for her diabetes insipidus, and she was started on daily aspirin and prednisone, along with pantoprazole and vitamin D for stomach and bone protection. A limited echocardiogram 8 days after VATS showed significant improvement in pericardial effusion (Fig. 3). She was discharged on colchicine, bumetanide and bactrim for prophylaxis against Pneumocystis jiroveci pneumonia. Before going home, a cardiac MRI was done to record the extension of the disease and help with future monitoring (Fig. 4). It was planned to start vemurafenib due to the discovery of the BRAF mutation.
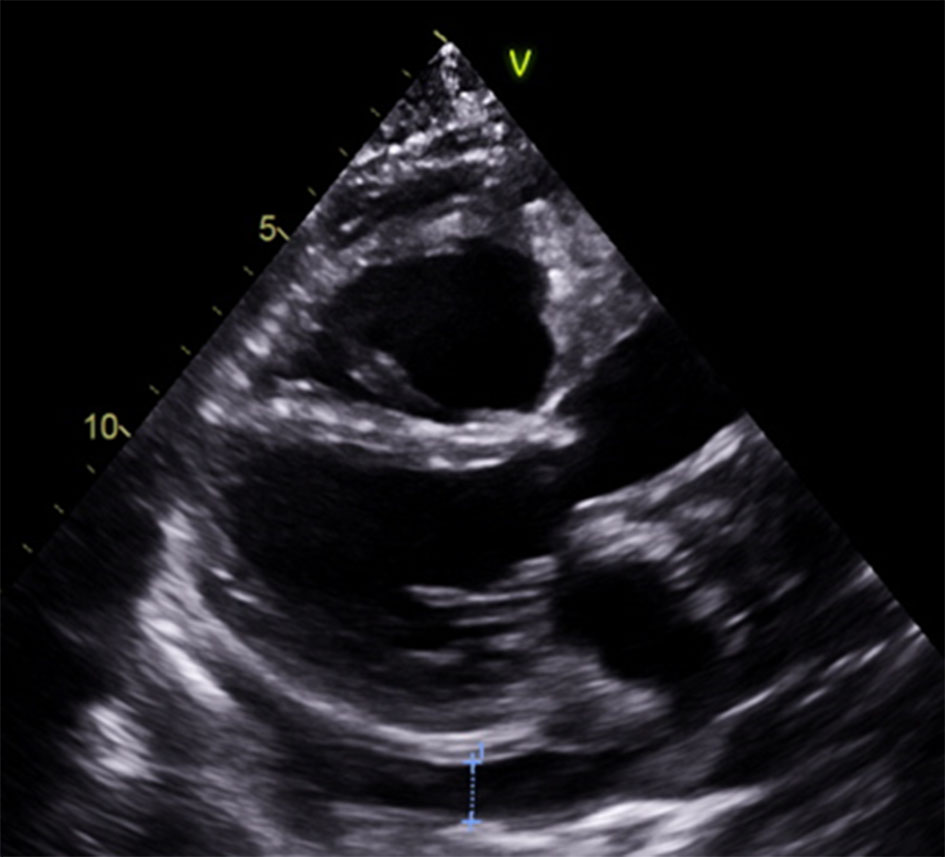 Click for large image | Figure 3. Reduction in pericardial effusion compared to Figure 1. |
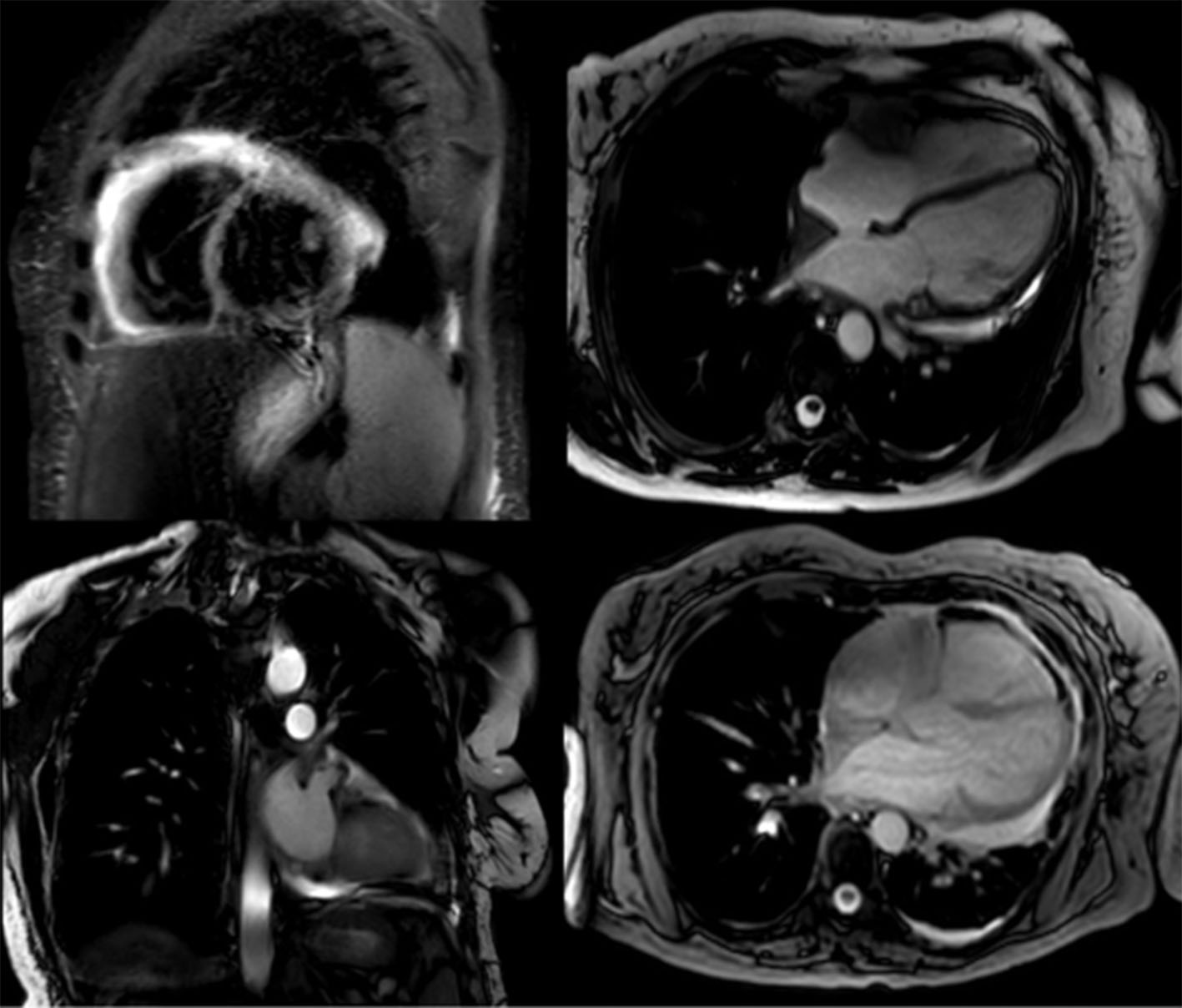 Click for large image | Figure 4. Cardiac MRI with very scarce effusion. MRI: magnetic resonance imaging. |
| Methods | ▴Top |
The process developed for the literature review included the following steps.
First, the relevant keywords were established. We conducted a broad search using the terms pericardial effusion and ECD.
Second, Scopus, PubMed, EMBASE, and Cochrane were searched for published articles through September 2019. The combinations of the terms used were: 1) SCOPUS, TITLE-ABS-KEY (“Erdheim-Chester Disease”) AND TITLE-ABS-KEY (“Pericardial Effusion”); 2) PUBMED, “Erdheim-Chester Disease” (Mesh) AND “Pericardial Effusion” (Mesh); 3) EMBASE, TITLE-ABS-KEY (“Erdheim-Chester Disease”) AND TITLE-ABS-KEY (“Pericardial Effusion”); and 4) COCHRANE, “Erdheim-Chester Disease” in Title Abstract Keyword AND “Pericardial Effusion” in Title Abstract Keyword (word variations have been searched). A total of 206 articles were found (84 from Scopus, 15 from PubMed, 107 from EMBASE and no articles found in Cochrane).
Third, article abstracts were reviewed for study eligibility, followed by reviews of full papers in cases in which eligibility was not clear from the abstract review alone. Another 18 full-text articles were excluded. Figure 5 shows the flow diagram according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [7].
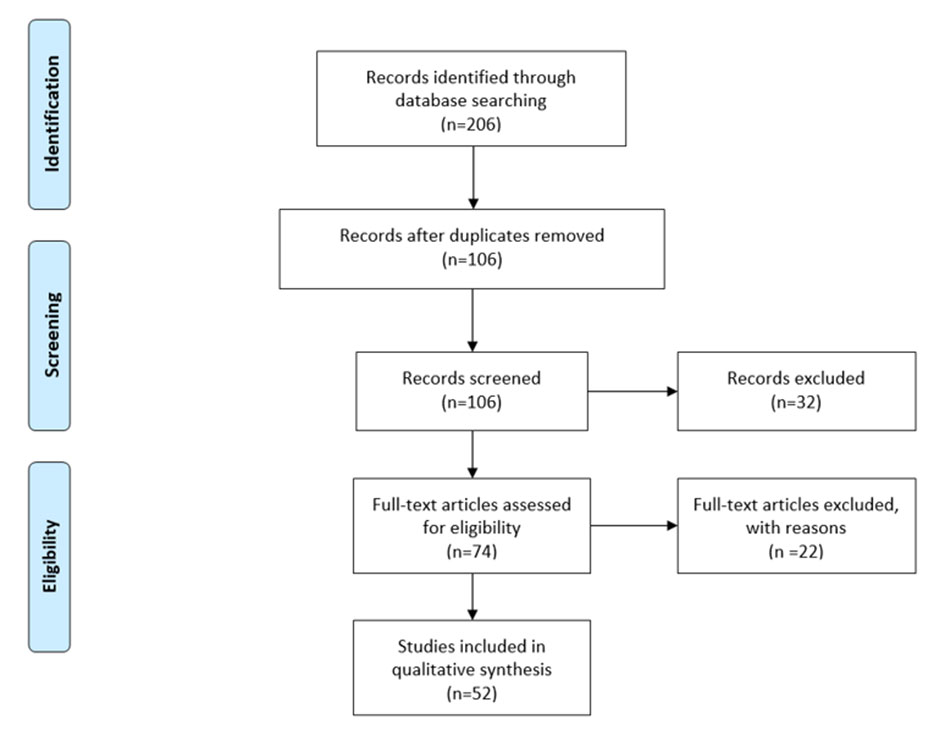 Click for large image | Figure 5. Flow diagram according to the PRISMA statement. PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses. |
Selection of studies
Articles were included if they consisted of presentation of pericardial effusion and ECD. Articles were excluded if they were primarily editorials, and solely described study protocols, letters, abstract of congresses or books. The results were summarized in tables.
| Results | ▴Top |
Fifty-two articles were included in the analysis. Table 1 shows the main characteristics of them. Most of the studies were case reports, eight were reviews and only three were original articles. Table 2 shows the case reports and case series in which patients with ECD presented with pericardial effusion [10-41]. The average age of the patients with ECD who presented with pericardial effusion was 56.64 ± 10.5. The majority of them were men (54.29%). Table 3 shows the interventions in patients reviewed. Almost 40% of the patients reviewed did not need any cardiologic procedure. A total of 30.3% of the patients received pericardiocentesis. Four received pleuro-pericardial window; three, pericardial window and two, pericardiectomy. The following procedures were performed in only one patient: mediastinoscopy, pericardial drainage, and sternotomy. One patient had pericardiocentesis and later received pericardiectomy, and another had pericardiocentesis done twice and later a pleuro-cardial window.
 Click to view | Table 1. Characteristics of Articles |
 Click to view | Table 2. Reports That Present Cases of Erdheim-Chester Disease With Pericardial Effusion |
 Click to view | Table 3. Interventions Performed in the Cases of Erdheim-Chester Disease With Pericardial Effusion |
| Discussion | ▴Top |
Approximately 75% of patients with ECD suffer cardiovascular manifestations and about 60% of them will perish due to cardiac complications [10]. Patients with ECD and cardiac involvement have a reduced response to treatment and a poor overall prognosis [42]. There are several types of cardiac injuries that can cause congestive heart failure, including myocardial infarction, thromboembolism, cardiac remodeling, valve dysfunction, ischemia, and peripheral edema among others. Pericardial infiltration is the most frequent cardiac manifestation of ECD [43] and is the one discussed in this article.
Pericardial involvement in ECD takes several forms and the symptoms are correlated with type, extent, and severity. Pericardial infiltration is easily discovered with computed tomography and transthoracic echocardiography. The pericardium may appear thickened with or without a fibrosis wrap. The accumulation of pericardial effusion can lead to cardiac tamponade. This can be relieved with pericardiocentesis, which has diagnostic and therapeutic purposes. Aggressive interventions may include placement of a pericardial window, pericardiectomy, and placement of a pericardial-abdominal shunt. Cytological examination of pericardial effusion can reveal mesothelial cells, histiocytes, and inflammatory cells. A histological examination of a pericardial sample may reveal the infiltration of foamy histiocytes [44]. In our revision, we found that the majority of the patients with pericardial infiltration needed a cardiovascular procedure. Pericardial infiltration can manifest as pericardial thickening and symptoms vary according to the degree of disease severity. Myocardial involvement occurs sequentially to the pericardial involvement and manifests as myocardial hypertrophy, easily diagnosed by the echocardiogram. Thickening can be found in the ventricles, atria, coronary sulci and interatrial septum [11].
To date, several treatments have been administered to patients with ECD in an attempt to achieve remission or at least stabilization. Currently, interferon-α provides the best management strategy, with a sustainable stabilization of the disease in most cases [45-48]. Interferon-α is administered in doses that vary from three million units three times per week to nine million units three times per week. Peginterferon-α-2a, the pegylated form of interferon-α, is an equivalent alternative to interferon-α. It is administered in doses that vary from 135 to 200 µg per week [45]. It should be noted that treatment with these agents is prolonged. Therefore, treatment tolerance should be considered fundamental in long-term treatment planning. Among the adverse effects of interferon-α are asthenia, myalgia, pruritus, thrombocytopenia, and depression [49].
The efficacy of interferon-α varies according to the different sites of disease involvement. A recent survey involving 24 patients treated with high doses of interferon-α (> 18 million units per week) or high doses of peginterferon-α-2a (> 185 µg per week) reported that they were effective in treating patients with severe ECD. The response to treatment was most prominent in the cutaneous foci of the disease, followed by foci in the central nervous system (CNS) and pituitary; foci of the lung and heart were more resistant to treatment [49]. Interferon-α treatment itself was identified as an independent predictor of survival. It is currently the only agent that has been shown to improve survival among patients with ECD. Cladribine is recommended as an alternative treatment for ECD. However, the literature contains significantly fewer data regarding this medication compared to interferon-α [45].
In the past, vemurafenib proved to be an effective therapeutic option in patients with melanoma-BRAF V600 mutation; the reason why this medication was tested in patients with ECD was a link found between this disease and a specific BRAF V600 mutation. A nonrandomized, open-label phase 2 clinical trial by Diamond et al showed long-term efficacy in patients with BRAF V600-mutant ECD [8]. Vemurafenib gained FDA approval in 2017 with an oral dose of 960 mg every 12 h. Common side effects include arthralgia, alopecia, maculopapular rash, fatigue, photosensitivity, uveitis, and prolonged QT interval. Skin cancer, anaphylaxis, drug reaction with eosinophilia and systemic symptoms (DRESS) syndrome, Stevens-Johnson syndrome, toxic epidermal necrolysis, hepatotoxicity, kidney failure, Dupuytren’s contracture and teratogenicity have been described as rare but severe side effects.
Conclusions
ECD is a rare non-Langerhans cell histiocytosis characterized by systemic xanthogranulomatous infiltration. We described the case of a female adult presenting with pericardial effusion. Pericardial infiltration is the most frequent cardiac manifestation of ECD and is the one discussed in this article. We found that the majority of the patients with pericardial infiltration needed a cardiovascular procedure.
Acknowledgments
We would like to express our sincere thanks to Mr. Breandan Richard Quinn for his help and assistance in the editing and proofreading of this article.
Financial Disclosure
This project was not supported by any grant or funding agencies.
Conflict of Interest
The authors declare that they have no competing interest.
Informed Consent
It was determined that informed consent was not necessary because data was obtained from medical records and anonymized.
Author Contributions
All authors contributed to manuscript writing and approved the final version. ASN, AAG, and JNM contributed to conception and design and data analysis and interpretation. AAG contributed to the collection and assembly of data and design and data analysis and interpretation. ASN, AAG, and JNM helped with data analysis and interpretation. All authors read and approved the final manuscript.
Data Availability
The data supporting the findings of this study are available from the corresponding author upon reasonable request.
| References | ▴Top |
- Unal E, Karcaaltincaba M, Akpinar E, Ariyurek OM. The imaging appearances of various pericardial disorders. Insights Imaging [Internet]. 2019;10(1):42.
doi pubmed - Nicolazzi MA, Carnicelli A, Fuorlo M, Favuzzi AMR, Landolfi R. Cardiovascular involvement in Erdheim-Chester disease. Medicine (Baltimore) [Internet]. 2015;94(43):e1365.
doi pubmed - Chester W. Lipoidgranulomatose. Virchows Arch Pathol Anat. 1930;279:561-602.
doi - Consensus guidelines for the diagnosis and clinical management of Erdheim-Chester disease. Blood Journal [Internet]. Available from: http://www.bloodjournal.org/content/early/2014/05/21/blood-2014-03-561381/tab-article-info.
- Maladie d'Erdheim-Chester - ScienceDirect [Internet]. Available from: https://www.sciencedirect.com/science/article/pii/S0248866314004755.
- Mazor RD, Manevich-Mazor M, Shoenfeld Y. Erdheim-Chester Disease: a comprehensive review of the literature. Orphanet J Rare Dis. 2013;8:137.
doi pubmed - Haroche J, Charlotte F, Arnaud L, von Deimling A, Helias-Rodzewicz Z, Hervier B, Cohen-Aubart F, et al. High prevalence of BRAF V600E mutations in Erdheim-Chester disease but not in other non-Langerhans cell histiocytoses. Blood. 2012;120(13):2700-2703.
doi pubmed - Diamond EL, Subbiah V, Lockhart AC, Blay JY, Puzanov I, Chau I, Raje NS, et al. Vemurafenib for BRAF V600-mutant Erdheim-Chester disease and langerhans cell histiocytosis: analysis of data from the histology-independent, phase 2, Open-label VE-BASKET study. JAMA Oncol. 2018;4(3):384-388.
doi pubmed - Al Bayati A, Plate T, Al Bayati M, Yan Y, Lavi ES, Rosenblatt JD. Dabrafenib and trametinib treatment for Erdheim-Chester disease with brain stem involvement. Mayo Clin Proc Innov Qual Outcomes. 2018;2(3):303-308.
doi pubmed - Alharthi MS, Calleja A, Panse P, Appleton C, Jaroszewski DE, Tazelaar HD, Mookadam F. Multimodality imaging showing complete cardiovascular involvement by Erdheim-Chester disease. Eur J Echocardiogr. 2010;11(7):E25.
doi pubmed - Costa I, Abdo ANR, Bittar CS, Fonseca SMR, Moraes A, Kalil Filho R, Pereira J, et al. Cardiovascular manifestations of Erdheim-Chester's disease: a case series. Arq Bras Cardiol. 2018;111(6):852-855.
doi pubmed - Andre M, Delevaux I, de Fraissinette B, Ponsonnaille J, Costes Chalret N, Wechsler B, Piette JC, et al. Two enlarged kidneys: a manifestation of Erdheim-Chester disease. Am J Nephrol. 2001;21(4):315-317.
doi pubmed - Curgunlu A, Karter Y, Ozturk A. Erdheim-Chester disease: a rare cause of paraplegia. Eur J Intern Med. 2003;14(1):53-55.
doi - Johnson MD, Aulino JP, Jagasia M, Mawn LA. Erdheim-chester disease mimicking multiple meningiomas syndrome. AJNR Am J Neuroradiol. 2004;25(1):134-137.
- Palotas A, Bogats G, Lazar M, Papos M, Matin K, Pavics L. An isotope-diagnostic approach to Erdheim-Chester's disease of the heart. J Cardiovasc Pharmacol Ther. 2006;11(4):274-279.
doi pubmed - Saboerali MD, Koolen MG, Noorduyn LA, van Delden OM, Bogaard HJ. Pleural thickening in a construction worker: it is not always mesothelioma. Neth J Med. 2006;64(3):88-90.
- Palotas A, Bogats G, Lazar M, Papos M, Matin K, Pavics L. Radiopharmaceutical diagnosis of Erdheim-Chester's disease. Nucl Med Commun. 2007;28(1):63-65.
doi pubmed - Kim MS, Kim CH, Choi SJ, Won CH, Chang SE, Lee MW, Choi JH, et al. Erdheim-chester disease. Ann Dermatol. 2010;22(4):439-443.
doi pubmed - Lutz SZ, Schmalzing M, Vogel-Claussen J, Adam P, May AE. [Recurrent pericardial effusion as first manifestation of Erdheim-Chester disease]. Dtsch Med Wochenschr. 2011;136(39):1952-1956.
doi pubmed - Vega J, Cisternas M, Bergoeing M, Espinosa R, Zapico A, Chadid P, Santamarina M. [Erdhei-Chester disease: report of one case]. Rev Med Chil. 2011;139(8):1054-1059.
doi pubmed - Egan A, Sorajja D, Jaroszewski D, Mookadam F. Erdheim-Chester disease: The role of video-assisted thoracoscopic surgery in diagnosing and treating cardiac involvement. Int J Surg Case Rep. 2012;3(3):107-110.
doi pubmed - Ota M, Sakamoto M, Sato K, Yoshida Y, Funakubo Asanuma Y, Akiyama Y, Yamakawa M, et al. Immunopathological analysis of Erdheim-Chester disease with massive ascites. Intern Med. 2012;51(19):2825-2830.
doi pubmed - Takeuchi T, Sato M, Sonomura T, Itakura T. Erdheim-Chester disease associated with intramedullary spinal cord lesion. Br J Radiol. 2012;85(1011):e62-64.
doi pubmed - Dagna L, Corti A, Langheim S, Guglielmi B, Cobelli FD, Doglioni C, et al. Tumor necrosis factor α as a master regulator of inflammation in Erdheim-Chester disease: rationale for the treatment of patients with infliximab. J Clin Oncol. 2012;(28):e286-e290.
doi pubmed - Dogan R, Aydin A, Temel H, Ozer N, Onder S, Hazırolan T, et al. Erdheim-Chester disease with cardiac involvement: a new case report. Turk Gogus Kalp Dama. 2013;21:127-132.
doi - Alexiou J, Klastersky J. Erdheim-Chester disease: a case report. Am J Case Rep. 2015;16:361-366.
doi pubmed - de Miguel Criado J, Aguilera Del Hoyo LF, Garcia Del Salto L, Cueva Perez E, Casado Cerrada J, Nieto Llanos S, Porro Fernandez JC, et al. Case 224: Cardiac involvement in Erdheim-Chester disease. Radiology. 2015;277(3):916-921.
doi pubmed - Mishra AK, Mani S, George AA, Sudarsanam TD. Recurrent pericardial effusion and tamponade in a patient with Erdheim-Chester disease (ECD). BMJ Case Rep. 2015;2015:bcr2015212483.
doi pubmed - Abdellateef EE, Abdelhai AR, Gawish HH, Abdulmonaem GA, Abdelbary EH, Ahmed AI. The first reported case of Erdheim-Chester disease in Egypt with bilateral exophthalmos, loss of vision, and multi-organ involvement in a young woman. Am J Case Rep. 2016;17:360-370.
doi pubmed - Azadeh N, Tazelaar HD, Gotway MB, Mookadam F, Fonseca R. Erdheim Chester Disease treated successfully with cladribine. Respir Med Case Rep. 2016;18:37-40.
doi pubmed - Mathis S, Godeneche G, Haroche J, Milin S, Julian A, Berthomet A, Baron C, et al. Long-term outcome of basilar stenosis in Erdheim-Chester disease: A case report. Medicine (Baltimore). 2016;95(36):e4813.
doi pubmed - Nakhleh A, Slobodin G, Elias N, Bejar J, Odeh M. Rapid progression to cardiac tamponade in Erdheim-Chester disease despite treatment with interferon alpha. Mod Rheumatol. 2016;26(4):621-624.
- Peric P, Antic B, Knezevic-Usaj S, Radic-Tasic O, Radovinovic-Tasic S, Vasic-Vilic J, Sekulovic L, et al. Successful treatment with cladribine of Erdheim-Chester disease with orbital and central nervous system involvement developing after treatment of Langerhans cell histiocytosis. Vojnosanit Pregl. 2016;73(1):83-87.
doi pubmed - Kyriakopoulou M, Decaux G, El Mourad M, Casado-Arroyo R. Acute cardiac tamponade in a 77-year-old italian woman with Erdheim-Chester disease. Eur J Case Rep Intern Med. 2016;3(7):000451.
doi pubmed - Bentel JM, Thomas MA, Rodgers JJ, Arooj M, Gray E, Allcock R, Fermoyle S, et al. Erdheim-Chester disease associated with a novel, complex BRAF p.Thr599_Val600delinsArgGlu mutation. BMJ Case Rep. 2017;2017:bcr-2017-219720.
doi pubmed - Chasseur P, Kyriakopoulou M, Vokaer B, Beukinga I, Casado Arroyo R, Cogan E, Couturier B. A cytologic diagnosis of BRAF(V600E) Erdheim-Chester disease on pericardial fluid. Acta Clin Belg. 2017;72(5):369-371.
doi pubmed - Franco-Palacios D, McDonald A, Aguillard RN, Berry A. An unusual case of interstitial lung disease in a patient with cardiopulmonary syndrome as the initial presentation of Erdheim-Chester disease. BMJ Case Rep. 2017;2017:bcr-2017-220659.
doi pubmed - Zhu P, Li N, Yu L, Miranda MN, Wang G, Duan Y. Erdheim-Chester disease with emperipolesis: a unique case involving the heart. Cancer Res Treat. 2017;49(2):553-558.
doi pubmed - Liersch J, Carlson JA, Schaller J. Histopathological and clinical findings in cutaneous manifestation of erdheim-chester disease and langerhans cell histiocytosis overlap syndrome associated with the BRAFV600E mutation. Am J Dermatopathol. 2017;39(7):493-503.
doi pubmed - Monmany J, Granell E, Lopez L, Domingo P. Resolved heart tamponade and controlled exophthalmos, facial pain and diabetes insipidus due to Erdheim-Chester disease. BMJ Case Rep. 2018;2018:bcr-2018-225224.
doi pubmed - Tomelleri A, Cavalli G, De Luca G, Campochiaro C, D'Aliberti T, Tresoldi M, Dagna L. Treating heart inflammation with interleukin-1 blockade in a case of Erdheim-Chester disease. Front Immunol. 2018;9:1233.
doi pubmed - Haroche J, Amoura Z, Charlotte F, Salvatierra J, Wechsler B, Graux C, Brousse N, et al. Imatinib mesylate for platelet-derived growth factor receptor-beta-positive Erdheim-Chester histiocytosis. Blood. 2008;111(11):5413-5415.
doi pubmed - Gupta A, Kelly B, McGuigan JE. Erdheim-Chester disease with prominent pericardial involvement: clinical, radiologic, and histologic findings. Am J Med Sci. 2002;324(2):96-100.
doi pubmed - Haroche J, Amoura Z, Dion E, Wechsler B, Costedoat-Chalumeau N, Cacoub P, Isnard R, et al. Cardiovascular involvement, an overlooked feature of Erdheim-Chester disease: report of 6 new cases and a literature review. Medicine (Baltimore). 2004;83(6):371-392.
doi pubmed - Arnaud L, Hervier B, Neel A, Hamidou MA, Kahn JE, Wechsler B, Perez-Pastor G, et al. CNS involvement and treatment with interferon-alpha are independent prognostic factors in Erdheim-Chester disease: a multicenter survival analysis of 53 patients. Blood. 2011;117(10):2778-2782.
doi pubmed - Haroche J, Amoura Z, Trad SG, Wechsler B, Cluzel P, Grenier PA, Piette JC. Variability in the efficacy of interferon-alpha in Erdheim-Chester disease by patient and site of involvement: results in eight patients. Arthritis Rheum. 2006;54(10):3330-3336.
doi pubmed - Braiteh F, Boxrud C, Esmaeli B, Kurzrock R. Successful treatment of Erdheim-Chester disease, a non-Langerhans-cell histiocytosis, with interferon-alpha. Blood. 2005;106(9):2992-2994.
doi pubmed - Suzuki HI, Hosoya N, Miyagawa K, Ota S, Nakashima H, Makita N, Kurokawa M. Erdheim-Chester disease: multisystem involvement and management with interferon-alpha. Leuk Res. 2010;34(1):e21-24.
doi pubmed - Hervier B, Arnaud L, Charlotte F, Wechsler B, Piette JC, Amoura Z, Haroche J. Treatment of Erdheim-Chester disease with long-term high-dose interferon-alpha. Semin Arthritis Rheum. 2012;41(6):907-913.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Cardiology Research is published by Elmer Press Inc.


